History
The clinical history and presentation of human papillomavirus (HPV) infection vary according to the anatomic area involved. The predilection of certain genotypes of virus to infect certain epidermal sites largely determines areas of involvement.
Genital infection manifests as a warty lesion on the genital or anal area, although these warts are often not initially recognized. Cervical infection generally goes unnoticed and is discovered during cervical examination or Papanicolaou (Pap) testing.
Anogenital warts
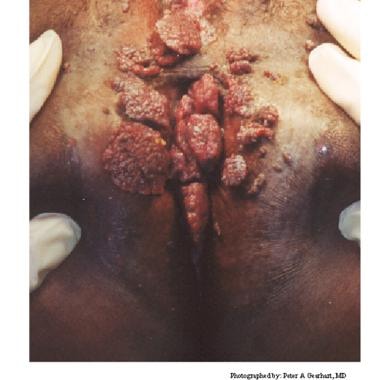
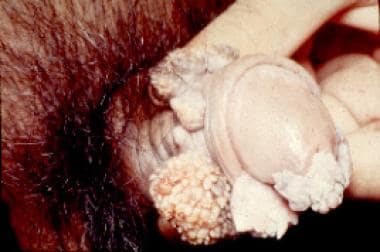
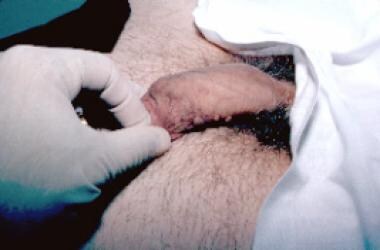
Condylomata acuminata are exophytic cauliflowerlike lesions that are usually found near moist surfaces. They may be observed in the perianal area, vaginal introitus, vagina, labia, and vulva. Genital warts may also be found on dry surfaces, such as the shaft of the penis.
Genital warts include smooth papular warts and keratotic warts, the latter of which resemble nongenital cutaneous warts because of their thickened bumpy surface.
Flat condylomata (squamous intraepithelial neoplasia) are the most common lesions of the cervix but may also develop on the vulva, anus, and male genitalia. They appear as white plaquelike growths.
An additional malignant variant is the giant condyloma, or Buschke-Löwenstein tumor, generally regarded as a verrucous carcinoma. These most often involve the glans penis, perianal area, and foreskin. In addition to their large cauliflower shape, they tend to form abscesses and fistulas and tend to invade locally.
Genital warts generally do not become clinically apparent until several months after inoculation with HPV. They follow a slow and indolent course and may develop by inoculation from opposing surfaces.
Condylomata acuminata are often asymptomatic. These lesions are generally not painful, but they can be associated with pruritus; bleeding may be observed if the lesions become confluent and are irritated by clothing. Most patients seek medical care when they notice lumps on the vulva, perianal area, or periclitoral area, or because they experience pruritus or occasional bleeding.
Cervical disease
Most cervical infections are either latent or subclinical and therefore asymptomatic. These infections are detected on Pap smear and are reported as either a low-grade squamous intraepithelial lesion (LGSIL) or a high-grade squamous intraepithelial lesion (HGSIL). Further examination with 3-5% acetic acid and colposcopy shows characteristic acetowhite changes and abnormal blood vessels indicative of HPV-triggered dysplasia.
Patients who have neglected to obtain annual Pap testing for several years or more and who have an HGSIL that has progressed to invasive cancer of the cervix may report vaginal bleeding between periods or after sexual intercourse, dyspareunia, and fullness in the pelvis.
Anal cancer
The most common presenting symptoms of squamous cell carcinoma (SCC) of the anus are rectal bleeding and sensation of a mass. These symptoms may be attributed mistakenly to hemorrhoids.
Fifty percent of men who are homosexual and have SCC of the anus have a history of anorectal warts; however, only 20% of women with SCC and men who are not homosexual have this history.
Nonanogenital mucosal disease
Oral warts represent infection of the oral mucosa. Although they are subtle and easily missed, they are fairly common. HPV types 6 and 11 have been isolated from nonanogenital mucosal surfaces. Warts have been discovered in the nares, mouth, larynx, and conjunctiva.
HPV types 6 and 11 are associated with respiratory papillomas that are probably the result of intrapartum transmission when the infant passes through the birth canal of a mother who is infected with HPV. However, isolated case reports exist of respiratory papillomatosis after cesarean delivery. Patients with laryngeal papillomas most frequently present with hoarseness at an average age of 3 years.
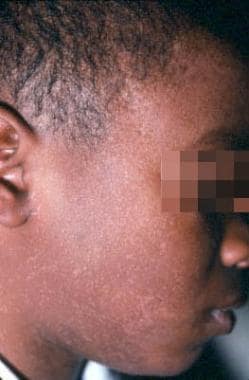
Focal epithelial hyperplasia (Heck disease) is a disseminated HPV infection of the oral mucosa most commonly associated with HPV 32 and HPV 13. This condition may have a family predilection.
Nongenital cutaneous HPV
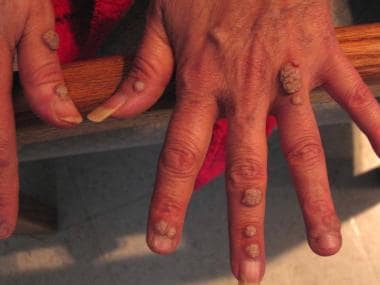
Common cutaneous warts (verruca vulgaris) generally appear on keratinized skin, presumably at the site of inoculation. Autoinoculation from a wart on one finger may cause the occurrence of warts on an adjacent finger or other skin surface (so-called kissing warts).
Common cutaneous warts appear as circumscribed, rough, hyperkeratotic papulonodules or plaques with irregular scaly surfaces and develop most often on the hands, fingers, feet, and knees. Such warts are frequently discovered when the patient notices changes in the skin. In general, they are asymptomatic, but they may be painful with application of pressure. Typically, they are benign and self-limited.
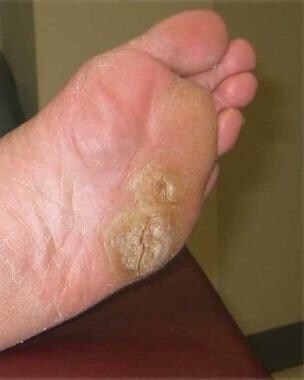
Palmoplantar warts appear on the acral surfaces of the feet and hands. They are notable for their thickness, which complicates treatment. Deep plantar warts occur most commonly as solitary lesions that may become black and painful before spontaneously regressing. They may contain small black “seeds,” which are thrombosed capillaries.
Warts that occur in people who handle meat and fish often have large cauliflowerlike plaques.
Flat warts (verruca plana) most often occur in groups of small plaques less than 5 mm in diameter on the face and hands. They often are not obvious but may induce significant disturbances of pigmentation. Regression usually occurs spontaneously after several years, and pruritus or erythema occurs several weeks before their disappearance.
Lloyd described Bowenoid papulosis as multicentric pigmented Bowen disease of the groin. It manifests as multiple, warty, red-brown papules in the anogenital region. These papules may coalesce.
Malignant conversion of skin lesions usually begins in the fourth and fifth decades of life. Premalignant lesions usually arise first on the forehead and other sun-exposed areas. The tumors are either benign papillomas and seborrheic keratoses or premalignant actinic keratoses and SCC.
Epidermodysplasia verruciformis
Epidermodysplasia verruciformis (EV) is an autosomal recessive familial trait that increases susceptibility to a subset of wart generally not observed in populations without EV. HPV genotypes associated with EV have been observed in patients who are immunosuppressed for organ transplantation or in patients with HIV infection. These individuals are at increased risk for skin cancer if not recognized and treated.
EV generally begins in childhood and can affect almost any area of the body. The warts are generally subtle and flat and may initially be mistaken for tinea versicolor. EV tumors are locally destructive. They develop slowly and have weak metastatic potential if no cocarcinogens, such as x-ray or ultraviolet B irradiation, are applied. Polymorphic, plane wart–like, and red-to-brownish plaques can be distributed widely over the skin. The lymph nodes and oral mucosa are not involved.
Physical Examination
The findings on physical examination depend on the tissues involved. They include a variety of cutaneous lesions with characteristic appearances (see History).
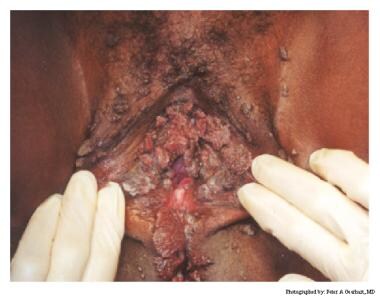
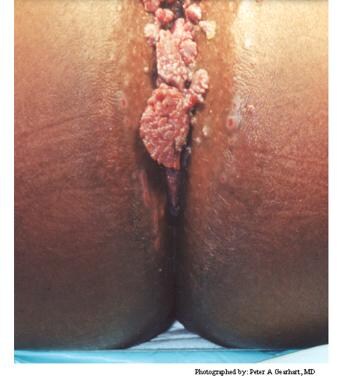
Typical condylomata are discrete, papillary, cauliflowerlike lesions that involve multiple sites on moist surfaces. Keratotic warts are often seen on dry surfaces like the labia majora. Warts vary in size and can form large, exophytic, cauliflowerlike masses (see the images below). Discrete papules 1-3 mm in size can present on the shaft of the penis. The growth can extend into the vagina, urethra, cervix, perirectal epithelium, anus, and rectum. Cervical intraepithelial lesions may be found upon examination of the cervix.
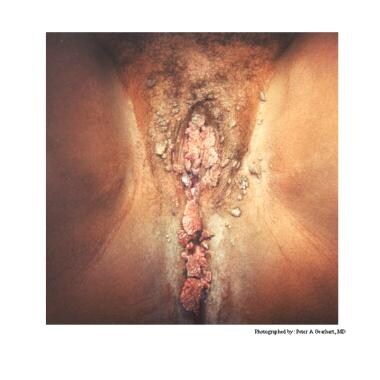 Human papillomavirus (HPV). Condyloma acuminatum in a patient with a history of an allograft renal transplant.
Human papillomavirus (HPV). Condyloma acuminatum in a patient with a history of an allograft renal transplant.
Subclinical infection is another common presentation of condyloma. Tiny, slightly raised areas can be felt or visualized on the vagina or cervix. These flat warts are best visualized by using 3-5% acetic acid and a colposcope. Areas infected with HPV appear acetowhite (ie, white when “painted” with the acetic acid). Often, a biopsy is needed to distinguish these lesions from cervical squamous intraepithelial lesions or vaginal intraepithelial lesions.
The sexual partner or partners of a woman with condylomata should be examined by a physician and treated if indicated. Often, the examination of the male fails to reveal any visible condylomata.
Complications
Some consider most common warts to be of only cosmetic concern and to cause few problems, unless their anatomic location induces mechanical problems. However, some patients may experience anxiety, guilt, and angst when they discover genital condyloma.
Plantar warts can disrupt ambulation because of their location. Laryngeal papillomas may disrupt breathing or speaking. Genital warts occasionally cause problems such as urethral obstruction. Condylomata acuminata can become extremely large, resulting in tissue breakdown or secondary infection. In the context of immune deficiency, such as HIV infection, the growth of warts due to HPV can be augmented, significantly enhancing the associated anatomic problems (see the image below).
However, disease complications can include progression to malignancy and transmission to other sexual contacts. In the setting of genital warts active during a pregnancy delivery, there is a small risk of laryngeal papillomatosis. Whereas the vast majority of genital HPV infections resolve spontaneously, persistent infection can lead to neoplastic changes. Cervical cancer is the second most common cause of morbidity and death in women in the United States. Malignancies such as Bowen tumors may also lead to morbidity and death.
Patients who develop condylomata acuminata have usually been exposed to low-risk HPV types such as HPV-6 and HPV-11. These HPV infections are associated with mild dysplasia that is often transient in nature. Many patients with mild dysplasia of the vulva, vagina, or cervix experience spontaneous regression of these lesions. Patients who are exposed to high-risk HPV types, such as HPV-16 or HPV-18, usually do not develop condylomata. These patients are at risk for high-grade dysplasia or anogenital carcinoma.
-
Human papillomavirus (HPV). Condyloma acuminatum in a patient with a history of an allograft renal transplant.
-
Human papillomavirus (HPV). Note the extensive labial involvement.
-
Human papillomavirus (HPV). Anal condyloma acuminatum.
-
Human papillomavirus (HPV). These condylomata extend to the anal verge.
-
Verrucous warts in patient with HIV infection.
-
Plantar warts.
-
Flat wart.
-
"Cauliflower" condyloma of penis.
-
Small papilloma on shaft of penis.
-
Human papillomavirus (HPV). Verruca vulgaris on the lateral border of the tongue exhibits the multiple, sharp-tipped, white, verrucous appearance, which is classic for this lesion in the oral cavity. Not all verrucae are so clinically diagnostic. Courtesy of Rose Yin Geist, DDS.
-
This is a verruca vulgaris of the anterior maxillary gingiva in a healthy young male. He had recently resolved a wart on his finger.
-
These small papillomas on the lateral tongue of a young woman showed histologic evidence of HPV in the form of extensive koilocytosis.
-
Human papillomavirus (HPV) lesion on the lingual frenum. Some patients with condyloma acuminatum present with multiple oral lesions. Courtesy of A.K. ElGeneidy, DDS.
-
Human papillomavirus (HPV). Condylomata on the lower lip, as well as other sites at the initial presentation. This presentation is unusually extensive. Multiple condylomata may be synchronous or metachronous. This patient did not present with genital condylomata. Courtesy of A.K. ElGeneidy, DDS.
-
Human papillomavirus (HPV). Heck disease in the buccal mucosa of a 7-year-old boy. Courtesy of Sheldon Mintz, DDS.
-
Human papillomavirus (HPV). Verrucae and papillomas appear as frondlike epithelial proliferations. Verrucae tend to be more keratinized with sharper projections than papillomas.
-
Human papillomavirus (HPV). Condyloma acuminatum generally has a papillary architecture and may microscopically resemble verruca vulgaris and papilloma (hematoxylin and eosin stain, original magnification X10). Courtesy of AK ElGeneidy, DDS.
-
Human papillomavirus (HPV). Koilocytes in the upper epithelium are a helpful, although not completely reliable, indication that a lesion is associated with human papillomavirus. Koilocytes display a dark small nucleus with clear cytoplasm (hematoxylin and eosin stain, original magnification X100). Courtesy of Sheldon Mintz, DDS.
-
Human papillomavirus (HPV). Condyloma acuminatum may show brisk mitotic activity, although oral condyloma acuminatum is not considered a premalignant lesion (hematoxylin and eosin stain, original magnification X40). Courtesy of A.K. ElGeneidy, DDS.
-
The left panel is transudation of serum antibodies to the site of human papillomavirus infection, and the right panel is exudation of serum antibodies to the site of human papillomavirus infection.
-
The figure shows proposed mechanisms used by the human papillomavirus vaccine to neutralize antibodies and protect against infection.
-
Figure showing how human papillomavirus penetrates the basal layer and eventually is released at the surface.