NORMAL PANCREAS ANATOMY
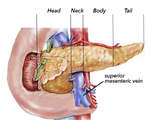
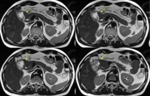
The pancreas is a coarsely lobulated gland typically 15–20 cm in length and located in the anterior pararenal space of the retroperitoneum.
The gland can be divided in four parts: head and uncinate process,
neck,
body and tail with the mesenteric vessels as a reference point [1-3] (Fig. 1).
- The pancreatic head is located within the curve of the duodenum,
to the right of the superior mesenteric vein.
The uncinate process is a prolongation of the caudal part of the head,
which is oriented toward the left.
- The pancreatic neck is the constricted portion to the left of the head and ventral to the superior mesenteric vein.
- The pancreatic body and tail lies to left of the superior mesenteric vein,
behind the lesser sac and the stomach.
The border between the body and tail is not clearly defined but can be determined using one-half of the distance between the neck and the end of the pancreas.
The duct of Wirsung unites with the common bile duct (CBD) and drains into the duodenum through the major papilla.
The duct of Santorini,
or accessory duct,
drains the anterior and superior portion of the head via the minor papilla.
Both pancreatic ducts communicate with each other near the neck of the pancreas forming a single remaining duct that runs through the center of the body and tail of the pancreas [4].
The distal CBD and duct of Wirsung traverse the sphincter of Oddi (which consists of three separate smooth muscles) to enter the duodenum.
In most cases (80%–90%) the CBD and duct of Wirsung unite within this sphincteric segment,
with the muscular wrap being 10 –15 mm in length.
The downstream ductal configuration most commonly (60%) encountered consists of a bifid configuration with patent ducts of Wirsung (ventral) and Santorini (dorsal) present.
Less common configurations include a rudimentary duct of Santorini (30%) and a dominant duct of Santorini (1%) [1,3].
For imaging of the pancreatic parenchyma,
various T2-weighted and T1-weighted sequences are used in transverse and coronal sections. The most important sequences are T1-weighted spoiled gradient echo with fat suppression and dynamic imaging before and after IV administration of gadolinium [5,6].
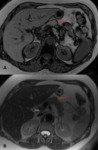
The normal pancreas has the highest signal intensity of the intra-abdominal organs on fat-suppressed T1WI (Fig. 2).
Its short T1 relaxation time has been attributed to the larger amount of aqueous protein within the glandular elements,
an abundance of endoplasmic reticulum of the protein-producing acinar cells,
and the high content of paramagnetic ions,
such as manganese,
within the pancreas. After IV gadolinium is administered,
the pancreas enhances maximally during the pancreatic arterial phase (15–20 sec).
During the later phases,
contrast material washes out and the pancreas becomes isointense relative to the liver [5,6].
Normal pancreatic parenchyma has a shorter T2 than most abdominal organs and,
therefore,
exhibits low-to-intermediate signal on T2WI. T2-weighted sequences are especially useful for the evaluation of the biliary tract and pancreatic duct. Magnetic resonance cholangiopancreatography (MRCP),
which is based on heavily T2-weighted sequences,
depicts fluid such as bile with high signal intensity. [5,6].
SIGNAL ALTERATIONS
1.
HEMOCHROMATOSIS
Iron overload (hemochromatosis) can be classified as primary or secondary.
-
Primary hemochromatosis (PH) is a recessive autosomal genetic disorder that alters a protein involved in the regulation of iron absorption,
which results in accumulation of iron in the parenchymal cells of the liver and other organs. Primary hemochromatosis is considered the most common genetic disease in the white population (rate of disease,
0.2%–0.5% for homozygous disease and as much as 10% for heterozygous disease). Iron is predominantly deposited in the liver, pancreas,
heart,
pituitary gland,
thyroid,
and synovium [7-10].
-
Secondary hemochromatosis (SH) is related with any other non-genetic cause of iron accumulation in the organs.
The causes of SH include: another cause of increased absorption (such as cirrhosis),
myelodysplastic syndrome,
anemias related to ineffective erythropoiesis (eg,
thalassemia),
and exogenous increase by ingestion,
parenteral infusion,
or multiple transfusions. Iron is predominantly deposited in the reticuloendothelial system (spleen and Kupffer cells) [7,11].
The distribution of iron accumulation differs between these two forms; therefore,
they can be distinguished by using imaging methods in the majority of cases. MRI is the most sensitive and specific imaging modality in the diagnosis of hemochromatosis.
The accumulation of iron ions in the tissues,
because of the superparamagnetic properties of the ions,
causes local distortion in the magnetic fields and relaxation of the spins,
which results in shortening of the longitudinal relaxation time (T1),
the transverse relaxation time (T2),
and particularly the transverse relaxation time as affected by magnetic field inhomogeneity (T2*). This effect causes a loss of signal intensity in the affected organs that is proportional to the iron deposition [7,12,13].
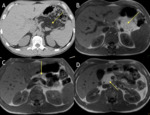
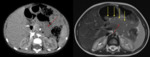
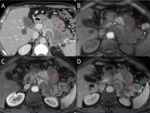
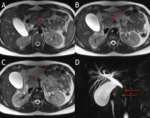
Pancreatic manifestations of haemochromatosis typically occur with PH,
as the organ is usually spared in the secondary form of the disease. Pancreatic involvement is uncommon in patients with PH without liver cirrhosis,
so that pancreatic signal intensity is often normal in precirrhotic patients with PH prior to the development of cirrhosis.
On the other hand the majority of cirrhotic patients with PH are diabetic,
the diabetes is secondary to both insulin resistance by hepatocytes and pancreatic beta cell damage from iron deposition,
so that the liver and pancreas of cirrhotic patients with PH typically have low signal intensity at MR. In cirrhotic patients with MR evidence of increased hepatic iron,
the cause of cirrhosis is less likely to be PH if pancreatic signal intensity is normal [8]. (Fig. 3).
2.
FATTY REPLACEMENT
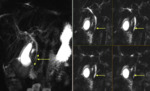
Pancreatic lipomatosis or fatty replacement of the pancreas is the most frequent pathologic finding in the adult pancreas [14].
Fatty replacement is distributed as patch reticular or multiple tiny nodules throughout the pancreas; however,
some investigators have found uneven fatty replacement of the pancreas,
which is seen as a focal fatty mass [15,16].
Focal fatty replacement of the pancreas is a rare benign disease [15].
It has been associated with a variety of diseases,
including obesity,
diabetes mellitus,
chronic pancreatitis,
steroid use,
hereditary pancreatitis,
obstruction of the pancreatic duct by calculus or tumor,
and cystic fibrosis. Fatty replacement occurs most frequently in anterior pancreatic head due to differences in fat content between ventral and dorsal pancreas.
It is not associated with mass effect or pancreatic ductal dilatation [17].
The posterior aspect of the head of the pancreas and the area around the CBD tend to be spared [18].
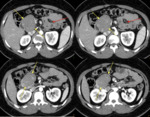
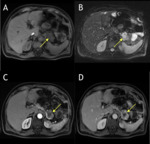
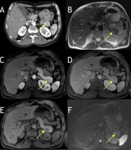
Focal fatty replacement may simulate a pancreatic mass such as a cystic neoplasm or other pancreatic tumor,
but also a hypoattenuating neoplasms in the pancreatic head should not be mistaken as focal fatty infiltration.[14].
MRI and CT are useful for confirming the presence of focal fatty infiltration and for differentiating neoplasm from fat deposition and focal areas of fat sparing. (Fig. 4).
Although unenhanced CT has an essential diagnostic role in depicting the same density as adjacent retroperitoneal fat with negative attenuation values,
a mild degree of focal fatty replacement of the pancreas cannot be diagnosed with CT alone.
Moreover,
contrast-enhanced CT cannot show a negative attenuation value of this focal fatty replacement because normal pancreatic parenchyma entrapped between fatty replacement areas exhibits contrast enhancement [14].
MRI may be helpful for confirming the presence of this type of focal fatty replacement of the pancreas,
and in several studies focal pancreatic fatty replacement has been diagnosed with MRI [19,20]. The T1-weighted in-phase sequence revealed the fatty infiltrated pancreas as having a signal intensity similar to or higher than that of the remainder of the pancreas.
This finding is in contradistinction to most pancreatic malignancies,
which appear as areas of low signal intensity on T1-weighted sequences [21].
Chemical shift MRI has an essential role in the correct diagnosis.
The loss of signal intensity on an opposed-phase T1-weighted gradient-echo image compared with a corresponding in-phase image establishes the lipid content of the focal abnormalities detected on prior CT examination and therefore excludes a diagnosis of pancreatic adenocarcinoma or neuroendocrine tumor (both of which do not contain lipid) [14,21].
2.
CYSTIC FIBROSIS
Cystic fibrosis (CF) is the most common lethal autosomal recessive disease in the white population [22,23,24]. Mutation of the CF transmembrane conductance regulator gene on chromosome 7 results in production of abnormally viscous mucus and secretions in the lungs of patients with CF. Although the main burden of disease is pulmonary,
CF is a multisystem disease with involvement of extrathoracic organs,
including the pancreas,
hepatobiliary system,
kidneys,
and gastrointestinal tract [25].
The pancreas is the abdominal organ most commonly involved in CF. Pancreatic involvement can result in exocrine and endocrine insufciency,
pancreatic atrophy,
fatty replacement,
or lipomatous pseudohypertrophy.
Acute and chronic pancreatitis,
pancreatic calcification,
cysts,
and cystosis also occur [25].
Inspissated secretions in the proximal pancreatic ducts result in obstruction and subsequent acinar disruption and replacement with fibrous tissue and fat.
Luminal obstruction and continued secretory capacity produce increased fluid pressure proximal to the obstruction that causes ducts to dilate and results in cyst formation and,
rarely,
progression to parenchymal cystic replacement [26]. The range of appearances includes complete replacement by fibrofatty tissue,
lipomatous pseudohypertrophy,
complete atrophy of the pancreas,
acute and chronic pancreatitis,
diffuse fibrosis with microcystic transformation,
and pancreatic cystosis [23,27-29].
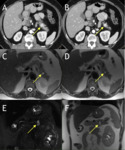
MRI and MRCP are useful,
noninvasive techniques for the assessment of pancreatic and hepatobiliary complications in CF. MRI can be used to effectively differentiate between fatty change,
which is characterized by hyperintensity at T1- and T2- weighted imaging,
and fibrotic change,
which is characterized by hypointensity at T1- and T2-weighted imaging [28].
Complete fatty replacement is the most common pancreatic finding at imaging in adult CF patients and the mean age of fatty replacement is 17 years [28,30-34].
At MRI,
the degree of fatty replacement will vary,
with increased signal intensity on T1-weighted images reflecting fatty change and intermixed low signal intensity on T1-weighted images representing fibrosis. (Fig. 5).
Pancreatic cysts are a relatively common finding in patients with CF and are typically quite small (1–3mm) but can reach a diameter of several centimeters.
Probably occur secondary to duct obstruction by inspissated secretions. Pancreatic cystosis is a rare manifestation of CF in which the pancreas is replaced by macroscopic epithelium- lined cysts of varying size that may cause mass effect within the abdomen [24,26,28].
The cysts are typically unilocular and have no internal septa or enhancement seen after intravenous injection of contrast material. At T1- and T2-weighted MRI,
the cysts are hypointense and hyperintense,
respectively,
compared with the background pancreatic tissue [35,36]. The imaging appearance can be similar to those of cystic pancreatic neoplasms,
von Hippel–Lindau disease,and autosomal dominant polycystic kidney disease [24].
CONGENITAL ANOMALIES
Congenital variants of the pancreas are seen in approximately 10% of the general population and include both malfusion variants such as pancreas divisum and malrotation variants such as annular pancreas and partial agenesis.
Most of these congenital variants are of limited clinical importance and are found incidentally at endoscopy,
surgery or autopsy,
and nowadays,
MRCP [37].
1.
PANCREAS DIVISUM
Pancreas divisum is the most common congenital anomaly of the pancreatic ductal system,
being reported in 4 -14% of the population at autopsy series,
3-8% at Endoscopic retrograde cholangiopancreatography (ERCP) and 9% at MRCP [37,38].
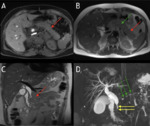
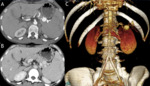
This anomaly results from nonfusion of the ventral and dorsal pancreatic ducts during embryologic life. The ventral duct (duct of Wirsung) drains only the ventral pancreatic anlage,
whereas the majority of the gland empties into the minor papilla through the dorsal duct (duct of Santorini) [3,39,40]. (Fig. 6).
In most cases,
there is no communication between the ventral and dorsal duct; however,
in some individuals,
there is a subtle communication remaining and in others the ventral duct is totally absent [40]. Focal dilation of the terminal portion of the dorsal duct,
also called Santorinicele,
is suggestive of relative obstruction at the minor papilla [3].
Pancreas divisum is usually asymptomatic,
but is more frequently seen in patients with chronic abdominal pain and idiopathic pancreatitis than in the general population. Although still debatable,
pancreas divisum is associated with acute and recurrent pancreatitis [40]. It is postulated that in pancreas divisum,
the duct of Santorini and the minor ampulla are too small to adequately drain the secretions produced by the pancreatic body and tail [41].
2.
ANNULAR PANCREAS
Annular pancreas is a rare congenital anomaly (1/2.000 people),
in which incomplete rotation of the ventral anlage leads to a band of pancreatic tissue surrounds the descending duodenum,
either completely or incompletely,
and is in continuity with the head of the pancreas [38,40,42,43].
The manifestation of annular pancreas in adults differs from that in children.
In children,
it usually manifests with symptoms resulting from gastric outlet obstruction or bile duct obstruction,
during the 1st year of life.
In adults,
it manifests with peptic ulcers,
duodenal obstruction,
or pancreatitis,
or in rare instances,
biliary obstruction; however,
approximately 50% of patients may be asymptomatic for life,
with the anomaly discovered incidentally [39,44-46].
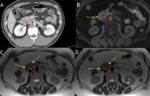
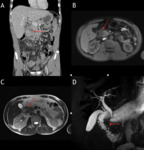
Annular pancreas can be diagnosed on the basis of CT and MRI findings that reveal normal pancreatic tissue,
with or without a small pancreatic duct,
encircling the descending duodenum [39,41,46].
MR show normal pancreatic tissue (with identical signal to normal pancreas) encircling the second portion of the duodenum (completely or partially) (Fig. 7). The descending duodenum is constricted and presents apparent wall thickening better seen on fat-suppressed T1 WI. MRCP may show the ventral component of the MPD encircling the duodenum,
which may drain into the intra-pancreatic CBD,
the duct of Wirsung,
or the duct of Santorini [42,47,48].
The encircling pancreatic tissue in these cases is not to be misinterpreted as lymph nodes,
duodenal carcinoma,
pancreatic ductal carcinoma, postbulbar peptic ulcer and multiple other causes of duodenal stenosis [42,49].
3. PANCREATIC AGENESIS
Total agenesis of the pancreas is extremely rare and is incompatible with life. Partial agenesis results from the absence of the ventral or dorsal anlagen.
Absence of the dorsal anlage is visualized as a short or truncated pancreas and can be partial or complete.
It may be seen as a solitary finding or in association with heterotaxia syndromes.
Partial agenesis of the dorsal pancreas is more common than agenesis of the ventral portion,
but complete agenesis of the dorsal pancreas is extremely rare [3,39,40].
Patients with agenesis of the dorsal pancreas often present with nonspecific abdominal pain,
which may or may not be caused by pancreatitis.
Many patients also have diabetes mellitus because most of the islet cells are located in the distal pancreas.
[3,39,47]. Pancreas hypoplasia may be associated with an increased risk of pancreatitis and polysplenia syndrome [39].
In patients with complete agenesis of the dorsal pancreas,
the neck,
body,
and tail of the pancreas,
the duct of Santorini,
and the minor duodenal papilla are all absent.
In cases of partial agenesis of the dorsal pancreas,
the size of the body of the pancreas varies,
there is a remnant of the duct of Santorini,
and the minor duodenal papilla is present.
These anomalies are better seen on cross-sectional T1 or T2 WI that show the absence of the dorsal pancreas associated with a short but normal pancreatic duct at MRCP [42,47 50].
(Fig. 8).
When agenesis of the dorsal pancreas is suspected,
it is critical to rule out a pancreatic neoplasm with upstream atrophy of the gland [3,39]. It also is crucial to distinguish agenesis of the pancreas from atrophy and lipomatous replacement of the pancreas secondary to chronic pancreatitis.
Dependent stomach and/or dependent intestine signs are useful imaging manifestations on cross-sectional imaging that allow differentiation of dorsal agenesis from distal lipomatosis,
this sign refers to the distal pancreatic bed getting filled by stomach or intestine which abut the splenic vein,
while,
in case of distal lipomatosis abundant fat tissue is observed anterior to the splenic vein [51].
4. PANCREATIC FETAL LOBULATIONS (PSEUDOMASSES)
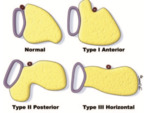
Alterations in the shape of the pancreas manifesting as pseudomasses can simulate pancreatic neoplasms.
Pancreatic head and neck lobulations are defined as outpouchings of the parenchyma more than 1.0 cm beyond the anterior superior pancreaticoduodenal artery.
These lobulations are present in approximately 34% of people and are classified in three main types: type I (anterior) seen in 10% of individuals; type II (posterior),
seen in 19%; and type III (horizontal or lateral),
seen in 5% [1,3,39,40]. (Fig. 9). Type I lobulations are often associated with a smaller uncinate process [1].
At imaging,
lobulations can be distinguished from tumor in that their characteristics are identical to those of the rest of the pancreas.
Relative to the surrounding pancreatic tissue,
lobulations are isoechoic at US, isoattenuating at CT,
and isointense at MRI,
and are isoenhancing following contrast material administration [1,41,47].
This is the key point that helps to differentiate this condition from a pancreatic neoplasm. (Fig. 10).
Another well-recognized pseudomass is a prominence of the anterior and superior surface of the pancreatic body to the left of the superior mesenteric vessels that abuts the posterior surface of the lesser omentum; this entity is known as “tuber omentale” or “omental tuberosity” and should be not misinterpreted as a pancreatic neoplasm or lymph node [1,3,39]. (Fig. 11).
5. ECTOPIC PANCREAS
Ectopic pancreas,
also referred to as heterotopic,
accessory,
or aberrant pancreas,
is defined as pancreatic tissue located outside of normal confines of pancreas and lacking anatomic and vascular continuity with the main body of the gland. [52,53]. The incidence in autopsy studies ranges between 0.6% and 15% [44]. Ectopic pancreatic tissue is usually seen in the submucosa of the gastric antrum (30%),
the proximal portion of the duodenum (30%),
the remaining duodenum (20%),
or other regions of the small bowel (20%) [40].
The majority of cases are asymptomatic and found incidentally.
However,
some patients with ectopic pancreas develop complications such as pancreatitis,
pseudocyst, insulinoma,
and pancreatic cancer,
which cause clinical symptoms such as abdominal pain,
gastrointestinal bleeding,
and obstruction [53].
Imaging plays a crucial role in the diagnosis of ectopic pancreas. There are no specific diagnostic features at CT to differentiate ectopic pancreas from other submucosal masses [47].
On CT,
ectopic pancreatic tissue is seen as a well-defined,
round or oval enhancing lesion in the antropyloric wall of the stomach,
or anywhere in the gastrointestinal tract.
Prominent mucosal enhancement overlying the lesion is a useful sign for differentiating ectopic pancreas from other submucosal lesions,
as enhancement is due to repeated inflammation of the mucosa by underlying pancreatic tissue. Another useful sign is the degree of enhancement of ectopic pancreas,
which is similar to that of normal pancreas in all phases [54].
On MR,
ectopic pancreatic tissue is isointense on all pulse sequences with main pancreas and enhancement is also similar to the normal pancreas in all phases [54]. MRCP is highly sensitive for identification of fluid containing structures such as an ectopic duct.
Ectopic pancreatic tissue can be diagnosed by identifying a duct from a mesenteric or bowel mass draining into the bowel lumen [55]. (Fig. 12). The ability of MRCP to depict an ectopic duct pathognomonic of ectopic pancreas may preclude the need for surgical excision to establish a definitive diagnosis in patients with this challenging condition [56].
Ectopic pancreas is often misdiagnosed,
as it may resemble other submucosal lesions,
such as GIST,
leiomyoma,
carcinoid,
neuroendocrine tumors and intramural metastasis,
on both imaging and during surgery. Knowledge of the various locations and imaging appearance of ectopic pancreas are important for the reporting radiologist in making an accurate diagnosis [54].
6.
ANOMALOUS PANCREATO-BILIARY JUNCTION
Anomalous union of the pancreatic and bile ducts is seen when the junction of the main pancreatic duct and CBD is outside of the duodenal wall,
resulting in a combined channel leading to the major papilla that measures more than 1.5 cm in length [40]. (Fig. 13). In this entity,
pancreatic secretions can reflux into the CBD and can result in the development of a bile duct cyst (type l) [57]. Conversely,
reflux of bile into the pancreatic duct can cause relapsing or chronic pancreatitis [39].
Reflux of pancreatic juice into the biliary tract might induce biliary tract damage and biliary carcinogenesis. Anomalous junction of the pancreaticobiliary ductal system has been reported in association with cholangiocarcinoma,
gallbladder carcinoma, choledocholithiasis,
and chronic pancreatitis [57].
Early diagnosis and early surgical treatment provide a good prognosis with few complications.
In addition,
successive follow-up is necessary for early detection of biliary tract malignancy,
especially in patients demonstrating postoperative complications [51].
8.
DISCREPANCY OF THE CALIBER OF THE MPD.
Discrepancy of the caliber of the MPD at the site of fusion of the dorsal and ventral ducts is normal variant that maybe confused for a site of stricturing.
The absence of dilatation of the proximal or upstream ductal system enables differentiation between those entities [3].
9.
BIFID TAIL OF THE PANCREAS
Duplication anomalies of the MPD are fairly common,
especially in the tail,
whereas parenchymal duplication is extremely rare [39]. Bifid pancreatic tail (pancreas bifidum),
or fish tail pancreas,
is a very rare congenital branching anomaly of the main pancreatic duct. It is identified as division of the pancreatic tail into separate dorsal and ventral buds. ( Fig. 14 ,
Fig. 15 ).
On MRCP or ERCP it manifests as duplication of the main pancreatic duct in the body of the pancreas. On cross-sectional imaging,
it is identified as division of the pancreatic tail into separate dorsal and ventral buds [51].
10. DUODENAL DIVERTICULA
Duodenal diverticula are common and are incidentally discovered at autopsy in 22% of cadavers.
A diverticulum may distort the periampullary anatomy and can even appear as a mass-like lesion.
On T2-weighted MRI,
duodenal diverticula may contain both high-signal-intensity areas,
related to the presence of fluid,
and low-signal- intensity areas,
related to the presence of gas [42]. (Fig. 16)
It may be difficult to distinguish duodenal diverticula on MRI if their content is purely fluid and they can be easily mistaken as cystic pancreatic neoplasms or inflammatory processes such as pseudocysts.
Careful scrutiny of images for evidence of subtle areas of decreased signal intensity that likely represent small amounts of intradiverticular gas or air– fluid levels should be made.
Visualization of decreased signal intensity or susceptibility artifact (blooming of low signal) on MRI sequences performed with increasing echo times in a periampullary cystic process supports the diagnosis of duodenal diverticulum [42]. In addition,
multidetector CT and coronal volume rendering may better delineate the thin,
intraluminal diverticulum sac wall and the plane of separation between the duodenum and pancreas [43].
IMAGING MIMICS OF PANCREATIC ADENOCARCINOMA
The accurate diagnosis of pancreatic adenocarcinoma is of critical importance,
because the disease has a high mortality and its treatment has substantial morbidity.
Several lines of evidence suggest that misdiagnosis,
either radiologically or pathologically,
may be relatively common [58].
1.
NEUROENDOCRINE TUMORS (NET).
Pancreatic NET account for 5% of pancreatic tumors and can be benign or malignant. Most cases are sporadic,
but association with syndromes such as multiple endocrine neoplasia type 1,
von Hippel–Lindau syndrome,
neurofibromatosis type 1,
and tuberous sclerosis has been observed [59].
The 5-year survival rate of 81% for pancreatic NET is significantly higher than that for pancreatic adenocarcinoma [58].
NETs are classified into functioning and nonfunctioning tumors.
Functioning tumors produce symptoms related to excessive hormone production.
They account for 15%–52% of all tumors and are subdivided according to the hormones they produce [59,60].
The hypervascularity of NET is fundamental to differentiate them from adenocarcinoma,
which is the most common solid pancreatic neoplasm,
and is mainly hypovascular.
At MRI, most NETs have low signal intensity on T1-weighted MRI and intermediate to high signal intensity on T2-weighted images.
The most distinctive feature of NETs is their behavior at contrast-enhanced imaging.
NETs have a rich vascular supply and therefore enhance avidly during the arterial phase,
enhancing more rapidly and intensely than the normal pancreas. Enhancement patterns include homogenous,
ring and/or diffusely heterogeneous immediately post-contrast administration.
Homogeneous enhancement is typical for small tumors less than 2 cm,
whereas larger lesions tend to show heterogeneous enhancement,
which can be ringlike.
Capturing this vascular blush is essential for the diagnosis of small tumors such as insulinoma,
which often do not distort the contour of the pancreas.
During the portal phase,
tumors may be either hyper-, iso-,
or hypoenhancing relative to the normal pancreas [59,61,62].
Some tumors can demonstrate heterogeneous and atypical delayed enhancement (best appreciated at portal phase imaging) and then can mimic pancreatic adenocarcinoma [59,61].
(Fig. 17, Fig. 18). Unlike adenocarcinoma,
pancreatic NETs are often well- circumscribed and duct obstruction is uncommon. In the same way,
calcifications (20% vs 2%),
tumor thrombus,
central necrosis and cystic degeneration are more common in NETs [59].
2. METASTASIS TO THE PANCREAS
Pancreatic metastases are rare,
with a reported incidence varying from 2 -11% in autopsy studies of patients with advanced malignancy. Metastases are most frequently from renal cell carcinoma (RCC) and lung carcinoma,
followed by breast carcinoma,
colorectal carcinoma,
and melanoma [63]. The time interval between the diagnosis of primary carcinoma and the detection of pancreatic metastases varies but is usually less than 3 years.
However,
metastases from RCC typically manifest 6–12 years (and as long as 20 years) after nephrectomy.
Three morphologic patterns of involvement are recognized: solitary (50%–70% of cases),
multifocal (5%–10%),
and diffuse (15%–44%) [63-66]. The majority of pancreatic metastasis appear as a large,
solitary mass with well-defined margins,
but cystic lesions may occur, Metastases do not show any predilection for a specific area of the pancreas [43].
Metastases typically have low signal intensity on T1- weighted MRI and high signal intensity on T2-weighted images.
At contrast-enhanced CT and MRI,
the appearances of pancreatic metastases closely resemble that of primary carcinoma [59].
The main differential consideration for pancreatic metastases from hypervascular primary tumors such as RCC is NET.
The imaging appearances of these two entities can be very similar.
Both manifest as hypervascular discrete masses that can exhibit cystic-necrotic degeneration.
However hypovascular metastases from (for example) the lung,
breast,
and colon are often seen,
and the main differential consideration for these lesions (in the absence of multiple metastases) is pancreatic adenocarcinoma [58,67]. (Fig. 19).
Of all tumors that metastasize to the pancreas,
lymphomas,
renal cell carcinomas,
and melanomas cause the most difficulty in the differentiation from primary pancreatic cancer because of their tendency to form solitary pancreatic lesions,
often without other identifiable metastatic sites. In general,
imaging factors that may favor metastatic disease include: multiplicity of lesions,
hypervascularity,
and imaging features consistent with a specific primary tumor,
such as hyperintensity of melanoma at T1-weighted MRI due to paramagnetic melanin content.
Clinical history of a known primary malignancy would help guide a differential diagnosis [43].
3.
PANCREATIC LYMPHOMA
Pancreatic lymphoma is most commonly a B-cell subtype of non-Hodgkin lymphoma and is classified as either primary or secondary. Secondary lymphoma is the dominant form and is the result of direct extension from peripancreatic lymphadenopathy. Primary pancreatic lymphoma (PPL) is rare. It is more common in middle-aged patients (mean,
55 years; range,
35–75 years) and in immunocompromised patients (eg,
patients with human immunodeficiency virus,
transplant recipients) [59].
The clinical presentation is nonspecific,
with the most common findings being abdominal pain (83% of cases),
a mass (58%),
and weight loss (50%). Obstructive jaundice (37%–42% of cases) and acute pancreatitis (12%) are less commonly seen [65]. PPL can present as an isolated mass mimicking pancreatic carcinoma. However,
unlike carcinomas,
PPL carries a better prognosis than adenocarcinoma because first-line treatment with chemotherapy is generally effective in producing long-term disease regression or remission , which is one reason why it is important to diagnose correctly [68].
A primary lesion of the pancreas may be difficult to distinguish from adjacent disease in the peripancreatic lymph nodes or duodenum. Anterior displacement of the pancreas or an intact fat plane between the pancreas and adjacent disease is a distinguishing feature. Two morphologic patterns of pancreatic lymphoma are recognized: a focal well-circumscribed form and a diffuse form. The focal form occurs in the pancreatic head in 80% of cases and has a mean size of 8 cm (range,
2–15 cm). It typically has uniform low attenuation at CT. At MR imaging,
it has low signal intensity on T1-weighted images and intermediate signal intensity on T2- weighted images (slightly higher signal intensity than the pancreas but lower signal intensity than fluid),
and shows faint contrast enhancement.
The diffuse form is in infiltrative,
leading to glandular enlargement and poor definition,
features that can simulate the appearance of acute pancreatitis.
This form has low signal intensity on T1- and T2-weighted MR images and shows homogeneous contrast enhancement,
although small foci of reduced or absent enhancement are sometimes seen [59,69]. (Fig. 20).
Several features of lymphoma may help distinguish it from adenocarcinoma [59,68,70]:
- A bulky localized tumor in the pancreatic head without significant MPD dilatation strengthens a diagnosis of pancreatic lymphoma over adenocarcinoma. The MPD is typically mildly dilated in lymphoma but grossly dilated in adenocarcinoma.
CBD dilatation is more common than MPD dilatation in lymphoma.
- Enlarged lymph nodes below the level of the renal vein.
- Invasive tumor growth that does not respect anatomic boundaries and that infiltrates retro- peritoneal or upper abdominal organs and the gastrointestinal tract.
- Lymphoma may encase vessels,
but exerts little mass effect and unlike adenocarcinoma does not typically invade vessels or produce thrombosis.
- Intratumoral calcification-necrosis is not a feature of lymphoma and may be helpful in its exclusion.
4.
GROOVE PANCREATITIS
Groove pancreatitis (GP) is a rare form of focal chronic pancreatitis affecting the “groove” between the superior aspect of the pancreatic head,
the duodenum,
and the CBD. The exact underlying cause of GP is unclear,
although a number of different theories exist: functional obstruction of the minor papilla or duct of Santorini,
increasingly viscous pancreatic secretions as a result of alcohol use or smoking,
Brunner gland hyperplasia resulting in stasis of pancreatic secretions in the dorsal pancreas,
heterotopic pancreas in the duodenum,
and peptic ulcer disease have all been suggested as potential contributing factors.
However,
a long history of alcohol abuse is thought to be the strongest association [71,
72].
There are two forms of GP: the segmental form,
which involves the pancreatic head with scar tissue in the pancreaticoduodenal groove; and the pure form,
which affects the groove but spares the pancreatic head. The segmental form,
which involves both the pancreatic head and the pancreaticoduodenal groove,
may resemble a focal mass at imaging,
thereby mimicking adenocarcinoma. GP is associated with smooth stricturing of the intrapancreatic portion of the CBD and wall thickening and cystic dysplasia of the duodenum [59].
The differential diagnosis of GP from pancreatic head adenocarcinoma has been reported to be difficult,
since specific discriminating imaging features are lacking,
and thus patients with GP may undergo unnecessary pancreatoduodenectomy for suspected pancreatic carcinoma [72].
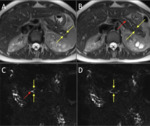
The most characteristic MR finding is a sheet-like mass between the head of the pancreas and the thickened duodenal wall associated with cystic changes in both the duodenal wall and pancreaticoduodenal groove.
The mass is hypointense on T1-weighted images compared to the hyperintensity of the rest of the gland and on T2-weighted images it appears bright in the edematous stage and progressively hypointense in the fibrotic stage [42]. (Fig. 21) The main pancreatic duct may appear normal in the pure form of GP,
but in the segmental form a stricture is usually seen within the pancreatic head associated with proximal dilatation.
Contrast-enhanced dynamic images show a delayed enhancement that reflects the fibrous nature of the tissue.
Most pancreatic adenocarcinomas are relatively hypovascular and may present a significant scirrhous component; thus,
contrast enhancement patterns can be similar in both conditions [42].
5.
AUTOIMMUNE PANCREATITIS
Autoimmune pancreatitis (AIP) is the pancreatic manifestation of IgG4- related sclerosing disease.
It is a distinct type of chronic pancreatitis and is characterized by abundant infiltration of IgG4-positive plasma cells and associated fibrosis that lead to organ dysfunction.
The involvement of pancreatic parenchyma by means of inflammation and fibrosis may be focal (most frequently occurring in the head of the gland) diffuse or multifocal [73].
Within the involved parenchyma,
the pancreatic ducts are usually stenotic,
and they sometimes have thickened walls [74]. In case of focal pancreatic involvement,
there is often an infiltrating mass-like parenchymal lesion,
that could be misinterpreted as pancreatic carcinoma.
This is because it is often associated with narrowing of the CBD [43]. AIP is an important mimic of pancreatic carcinoma; 3%–9% of patients who undergo resection for a presumed carcinoma have AIP [75],
therefore,
differentiating focal forms of AIP from pancreatic adenocarcinoma is important to avoid unnecessary surgery [76].
Numerous extrapancreatic organs,
such as the bile ducts,
gallbladder,
kidneys,
retroperitoneum,
mesentery,
thyroid,
lacrimal glands,
salivary glands,
lung,
mediastinum,
lymph nodes,
gastrointestinal tract and prostate may be involved,
either synchronously or metachronously [73].
Occasionally,
AIP is associated with other autoimmune disorders,
such as chronic inflammatory bowel disease,
primary sclerosing cholangitis, Sjogren syndrome,
retroperitoneal fibrosis,
diabetes mellitus,
systemic lupus erythematosus,
vasculitis,
or thyroiditis [76].
Imaging findings play a major role in the diagnosis of AIP,
and an awareness of its extrapancreatic findings is critical for accurate diagnosis. Focal disease is less common than diffuse disease and manifests as a focal mass,
often with involvement of the pancreatic head,
an appearance that may mimic that of pancreatic malignancy.
Focal disease tends to be relatively well demarcated,
and,
when present,
upstream dilatation of the main pancreatic duct is typically milder than in patients with pancreatic carcinoma.
The affected area of the pancreas typically appears hypoechoic at US,
hypoattenuating at CT,
mildly hyperintense at T2-weighted MRI,
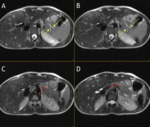
and hypointense at T1-weighted MRI. (Fig. 22) At contrast-enhanced CT and MRI,
decreased enhancement often is seen in the early phase,
and moderate delayed enhancement is seen in the late phase,
a finding likely due to fibrosis.
The presence of a capsule-like rim or “halo” is common in patients with AIP and is believed to represent fluid,
phlegmon,
or fibrous tissue. When the pancreatic head is involved,
narrowing of the intrapancreatic portion of the CBD without significant upstream dilatation typically is seen [42,73]. The periductal infiltrate can extend to the suprapancreatic segment of the CBD,
with a pattern resembling that of primary sclerosing cholangitis [77].
The main pancreatic duct typically is narrow and irregular in the segments affected by AIP, a finding that may be demonstrated at ERCP or MRCP [73].
Extrapancreatic abnormalities associated with IgG4-related systemic disease may be seen,
including salivary gland uptake on positron emission tomography (PET),
retroperitoneal fibrosis, periaortitis,
inflammatory renal pseudo-tumors,
and autoimmune cholangiopathy.
In addition to these extrapancreatic features,
the lack of pancreatic duct dilatation is a key feature that distinguishes AIP from adenocarcinoma [60]. In the context of a coexisting autoimmune disease such as primary sclerosing cholangitis or ulcerative colitis,
suspecting this diagnosis will help avoid unnecessary surgical intervention.
6.
FOCAL CHRONIC PANCREATITIS
Chronic pancreatitis can present as a focal inflammatory mass,
often in the pancreatic head,
thereby mimicking adenocarcinoma. This manifestation accounts for 5%–10% of pancreatectomies for presumed malignancy. Discrimination between focal pancreatitis and adenocarcinoma is difficult clinically,
radiologically,
and even histologically.
Both conditions can present with abdominal pain,
obstructive jaundice,
and weight loss.
Imaging results are confusing because inflammation can coexist with adenocarcinoma and adenocarcinoma may arise in long-standing chronic pancreatitis [59].
In general,
adenocarcinoma and focal pancreatitis are hypoechoic at US,
are hypoattenuating at CT,
and have the same signal intensity at T1-WI and T2-WI images .
The double duct sign,
ductal strictures,
infiltration of adjacent fat,
arterial encasement,
and peripancreatic venous obstruction may be present in both entities.
Features that favor a diagnosis of focal pancreatitis include nondilated or smoothly tapering pancreatic and bile ducts coursing through the mass (“duct penetrating sign”),
irregularity of the pancreatic duct,
and the presence of pancreatic calcifications.
Features that favor a diagnosis of adenocarcinoma include abrupt interruption of a smoothly dilated pancreatic duct and upstream pancreatic gland atrophy.
A high ratio of duct caliber to pancreatic gland width is a recognized indicator of adenocarcinoma.
Focal pancreatitis tends to have limited atrophy and nonabrupt gradual narrowing of the dilated pancreatic duct or bile duct [58,59,78,79].
However,
pancreatic adenocarcinoma can be superimposed on patients with chronic pancreatitis and therefore specific findings of chronic pancreatitis may not be sufficient to exclude pancreatic malignancy [78].
The two entities could not be distinguished on the basis of time or degree of enhancement,
both presented hypointense masses on T1-weighted imaging with progressive delayed enhancement compared to the rest of pancreas [80]. A mass causing upstream chronic pancreatitis can sometimes be detected on early phase dynamic gadolinium- enhanced images.
The cancer is seen as a focal hypointense mass relative to the hypoenhancing region of chronic pancreatitis on early gadolinium-enhanced images.
In addition,
the duct dilation abruptly terminates at the mass [78].
Due to clinical and imaging overlap between chronic pancreatitis and pancreatic adenocarcinoma,
a percutaneous CT- or EUS-guided FNAB may be required if the distinction can- not be made [78].
IMAGING MIMICS OF PANCREATIC NET
Pancreatic NET have a distinct appearance,
typically characterized by a well-defined hypervascular mass best visualized on arterial phase images.
However,
a number of other lesions can mimic pancreatic NET.
1.
INTRAPANCREATIC ACCESSORY SPLEEN
Accessory spleens represent a congenital anomaly arising from aberrant splenic embryologic fusion,
resulting in the ectopic deposition of normal splenic tissue in abnormal sites.
It is found in approximately 10 -30% of the population,
with only one out of six cases (16%) occurring in the pancreatic tail. Because of their relatively high density and enhancement,
intrapancreatic accessory spleen (IPAS) can be easily mistaken for a primary pancreatic mass,
particularly a NET [81-83].
On CT and MRI,
IPAS is most often a small (1–3 cm),
well-defined lesion that matches the density and intensity of the spleen on all contrast phases and remains stable over consecutive imaging.
Because the spleen is generally more dense than the pancreas during all dynamic CT phases (arterial,
pancreatic,
portal),
IPAS generally shows greater enhancement than does the pancreas.
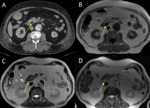
Relative to the pancreas,
IPAS (like the spleen) has lower signal intensity on T1-weighted MRI and higher signal intensity on T2-weighted images. The characteristic arciform splenic enhancement pattern seen during the arterial phase (secondary to perfusion differences between red and white pulp) is also seen in IPAS.
This is a useful criterion for differentiating IPAS from hypervascular neoplasms such as NETs and metastases [59]. (Fig. 23).
Technetium 99m (99mTc) sulfur colloid scintigraphy and 99mTc heat-damaged red blood cell (HDRBC) scintigraphy may be used to differentiate splenic from pancreatic tissue [42,59].
2.
SEROUS CYSTADENOMA
Serous cystadenoma (SCA) of the pancreas (or microcystic adenoma) is an uncommon type of benign cystic pancreatic neoplasm.
usually presents in middle age to elderly patients (60-80 years of age) [44]. The morphologic patterns of SCA can be classified as polycystic,
honeycomb,
and oligocystic [84].
- The polycystic pattern is the most common appearance of SCA; it occurs in approximately 70% of cases.
This pattern consists of a bosselated (marked by numerous rounded protuberances) collection of cysts that usually number more than six.
Each cyst may range from a few millimeters to 2 cm in size. This pattern shows two important morphologic features: external lobulation and a central scar.
Intraductal papillary mucinous neoplasm,
branch duct type,
and mucinous cystic neoplasm are cystic neoplasms that can have a polycystic appearance similar to that of SCAs.
- The honeycomb pattern is seen in approximately 20% of cases.
This pattern consists of numerous tiny cysts that mimic a honeycomb or a sponge.
These tiny cysts may be poorly depicted as individual cysts on CT or MRI.
On unenhanced CT,
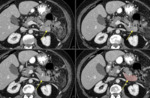
the honeycomb pattern may appear as a well-marginated lesion with soft-tissue or mixed attenuation, Such lesions may show moderate to strong enhancement and mimics pancreatic NET. MRI can be helpful for patients with a suspicious honeycomb pattern of SCA on CT. On MRI,
SCA can be seen as numerous discrete foci with bright signal intensities on the T2-weighted images. (Fig. 24).
- The macrocystic or oligocystic pattern is relatively uncommon.
Certain subtypes of serous tumors can have a very different appearance and mimics pancreatic NET:
- Extremely microcystic SCAs: are rare (5 %) and may resemble a solid lesion at conventional US and CT (pseudosolid SCAs).
After contrast medium administration,
these forms may resemble a hypervascular solid lesion,
owing to the homogeneous hyperenhancement of the extremely compacted internal septa.
The true cystic nature of the lesion can be demonstrated at MRI,
with a typical hyperintense signal on T2-weighted images [85].
- Solid serous adenoma: a extremely rare variant of SCA without an appreciable cystic component on histologic analysis,
appears completely solid on CT. These masses cannot be differentiated from pancreatic NET on CT. The utility of MRI for those serous tumors that are truly histologically solid is less certain [81].
3.
HYPERVASCULAR METASTASES
As previously mentioned,
metastases to the pancreas are rare. The appearance of most pancreatic metastases is not particularly specific,
although renal cell carcinoma metastases are usually highly vascular and can resemble NET.
In these cases,
a history of a primary malignancy (particularly renal cell carcinoma) should strongly raise the possibility of a pancreatic metastasis [81].
5.
VASCULAR LESIONS
Splenic artery aneurysms are estimated to occur in 0.1 to 10.4% of the general population and have a strong female predominance,
with a female to male ratio of 4:1.
Splenic artery aneurysms are defined by diameter greater than 1 cm.
It lesions may be mistaken for pancreatic NET when proper CT technique is not performed,
including high spatial resolution,
arterial and venous phase acquisitions,
as well as 2D multiplanar reconstructions and 3D rendering.
A single phase acquisition without narrow reconstruction sections can make the distinction of the two entities challenging [86]. Similar to aneurysms elsewhere the aneurysm follows the adjacent native vessel enhancement in all phases (Fig. 25).
Peripheral calcification can occasionally be seen [87].
Abnormalities such as aberrant vessels or pseudoaneurysms secondary to complicated pancreatitis or vascular surgical anastomoses may mimic a mass at routine imaging,
especially if the lumen is completely thrombosed and fails to enhance.
Venous structures may also be mistaken for pancreatic masses.
Examples include an unenhanced portal vein at true arterial phase imaging and poorly enhanced collateral vessels in thromboses of the portal vein or inferior vena cava [43].