Abstract
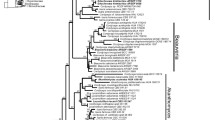
Data on morphology, anatomy, secondary chemistry, ecology, as well as phylogenetic reconstructions based on ITS/5.8S and mtSSU sequence data suggest that “Flavocetraria” minuscula should be transferred to a new monotypic genus Cladocetraria, phylogenetically related to “Cetraria” obtusata and Cetrariella. Cladocetraria minuscula is morphologically similar to Flavocetraria cucullata especially in miniature forms, but differs from this species in having helmet-shaped tips wrapped inside (up-turned) with inconspicuous white pruina, marginal pseudocyphellae in the form of a dotted line, and bacilliform uniformly thickened conidia. Differences from morphologically and phylogenetically related species are discussed. Chemical examination of Cladocetraria minuscula using a liquid chromatography–mass spectrometry assay revealed as previously reported usnic and protolichesterinic acids, as allo-protolichesterinic and lichesterinic acids reported for C. minuscula for the first time. According to presented phylogeny, Flavocetraria cucullata and F. nivalis are only distantly related and belong to different clades within the “Nephromopsis” branch, and must belong to different genera. In this regard, “Flavocetraria” nivalis should be transferred to the monotypic genus Foveolaria.





Similar content being viewed by others
References
Acharius E (1803) Methodus lichenum. Stockholm
Ashkenazy H, Sela I, Karin EL, Landan G, Pupko T (2019) Multiple sequence alignment averaging improves phylogeny reconstruction. Syst Biol 68:117–130. https://doi.org/10.1093/sysbio/syy036
Amo de Paz G, Cubas P, Divakar PK, Lumbsch HT, Crespo A (2011) Origin and diversification of major clades in parmelioid lichens (Parmeliaceae, Ascomycota) during the Paleogene inferred by Bayesian analysis. PLoS ONE 6:e28161. https://doi.org/10.1371/journal.pone.0028161
Blanco O, Crespo A, Elix JA, Hawksworth DL, Lumbsch HT (2004) A molecular phylogeny and a new classification of parmelioid lichens containing Xanthoparmelia-type lichenan (Ascomycota: Lecanorales). Taxon 53:959–975. https://doi.org/10.2307/4135563
Blanco O, Crespo A, Ree RH, Lumbsch HT (2006) Major clades of parmelioid lichens (Parmeliaceae, Ascomycota) and the evolution of their morphological and chemical diversity. Molec Phylogen Evol 39:52–69. https://doi.org/10.1016/j.ympev.2005.12.015
Chang JM, Floden EW, Herrero J, Gascuel O, Tommaso PD, Notredame C (2019) Incorporating alignment uncertainty into Felsenstein’s phylogenetic bootstrap to improve its reliability. Bioinformatics 37:1506–1514. https://doi.org/10.1093/bioinformatics/btz082
Chesnokov SV, Konoreva LA, Yatsyna AP, Andreev MP, Poryadina LN, Vondrák J, Himelbrant DE (2016) New and rare lichens from Republic of Sakha (Yakutia). II. Bull Tver State Univ Ser Biol Ecol 4:219–240 (In Russian)
Crespo A, Lumbsch HT, Mattsson J-E, Blanco O, Divakar PK, Articus K, Wiklund E, Bawingan PA, Wedin M (2007) Testing morphology-based hypotheses of phylogenetic relationships in Parmeliaceae (Ascomycota) using three ribosomal markers and the nuclear RPB1 gene. Molec Phylogen Evol 44:812–824. https://doi.org/10.1016/j.ympev.2006.11.029
Crespo A, Kauff F, Divakar PK, Prado R, Pérez-Ortega S, Paz GA, Ferencova Z, Blanco O, Roca-Valiente B, Núñez-Zapata J et al (2010) Phylogenetic generic classification of parmelioid lichens (Parmeliaceae, Ascomycota) based on molecular, morphological and chemical evidence. Taxon 59:1735–1753. https://doi.org/10.1002/tax.596008
Culberson CF, Kristinsson H (1970) A standardized method for the identification of lichen products. J Chromatogr 46:85–93. https://doi.org/10.1016/S0021-9673(00)83967-9
Del-Prado R, Cubas P, Lumbsch HT, Divakar PK, Blanco O, Amo de Paz G, Molina MC, Crespo A (2010) Genetic distances within and among species in monophyletic lineages of Parmeliaceae (Ascomycota) as a tool for taxon delimitation. Molec Phylogen Evol 56:125–133. https://doi.org/10.1016/j.ympev.2010.04.014
Divakar PK, Del-Prado R, Lumbsch HT, Wedin M, Esslinger TL, Leavitt SD, Crespo A (2012) Diversification of the newly recognized lichen-forming fungal lineage Montanelia (Parmeliaceae, Ascomycota) and its relation to key geological and climatic events. Amer J Bot 99:2014–2026. https://doi.org/10.3732/ajb.1200258
Divakar PK, Crespo A, Kraichak E, Leavitt SD, Singh G, Schmitt I, Lumbsch HT (2017) Using a temporal phylogenetic method to harmonize family- and genus-level classification in the largest clade of lichen-forming fungi. Fungal Diversity 84:101–117. https://doi.org/10.1007/s13225-017-0379-z
Elenkin AA, Savicz VP (1911) Enumeratio lichenum in Sibiria orientali a cl. I. Sczegolev anno 1903 lectorum. Trudy Bot Muz Imp Akad Nauk 8:26–49 (In Russian)
Gardes M, Bruns TD (1993) ITS primers with enhanced specificity for basidiomycetes – application to the identification of mycorrhizae and rusts. Molec Ecol 2:113–118. https://doi.org/10.1111/j.1365-294X.1993.tb00005.x
Guzow-Krzemińska B, Węgrzyn G (2000) Potential use of restriction analysis of PCR-amplified DNA fragments in taxonomy of lichens. Mycotaxon 76:305–313
Himelbrant DE, Stepanchikova IS, Kuznetsova ES (2014) Lichens. In: Neshataeva VY (ed) Vegetation cover of volcanic plateaus of Central Kamchatka. KMK Scientific Partnership, Moscow, pp 121–164 (In Russian)
Himelbrant DE, Stepanchikova IS, Ahti T, Neshataeva VYu (2019) The first lichenological survey in Koryakia (Northern Kamchatka, Russia)—the last unexplored part of Beringia. Novosti Sist Nizsh Rast 53:107–142. https://doi.org/10.31111/nsnr/2019.53.1.107
Jorgensen PM, James PW, Jarvis CE (1994) Linnaean lichen names and their typifications. Bot J Linn Soc 115:261–405. https://doi.org/10.1111/j.1095-8339.1994.tb01784.x
Kärnefelt I (1979) The brown fruticose species of Cetraria. Opera Bot 46:1–150
Kärnefelt I, Mattsson J-E, Thell A (1993) The lichen genera Arctocetraria, Cetraria and Cetrariella (Parmeliaceae) and their presumed evolutionary affinities. Bryologist 96:394–404. https://doi.org/10.2307/3243869
Kärnefelt I, Thell A, Randlane T, Saag A (1994) The genus Flavocetraria Kärnefelt & Tell (Parmeliaceae, Ascomycotina) and its affinities. Acta Bot Fenn 150:79–86
Katoh K, Kuma K, Tohand H, Miyata T (2005) MAFFT version 5: improvement in accuracy of multiple sequence alignment. Nucl Acids Res 33:511–518. https://doi.org/10.1093/nar/gki198
Kraichak E, Divakar PK, Crespo A, Leavitt SD, Nelsen MP, Lücking R, Lumbsch HT (2015) A tale of two hyper-diversities: diversification dynamics of the two largest families of lichenized fungi. Sci Rep 5:10028. https://doi.org/10.1038/srep10028
Kroken S, Taylor JW (2001) A gene genealogical approach to recognize phylogenetic species boundaries in the lichenized fungus Letharia. Mycologia 93:38–53. https://doi.org/10.1080/00275514.2001.12061278
Kurokawa S, Lai M (1991) Allocetraria, a new lichen genus in the Parmeliaceae. Bull Natl Sci Mus Tokyo B 17:59–65
Lanfear R, Calcott B, Ho SYW, Guindon S (2012) PartitionFinder: combined selection of partitioning schemes and substitution models for phylogenetic analyses. Molec Biol Evol 29:1695–1701. https://doi.org/10.1093/molbev/mss020
Larget B, Simon D (1999) Markov chain Monte Carlo algorithms for the Bayesian analysis of phylogenetic trees. Molec Biol Evol 16:750–759. https://doi.org/10.1093/oxfordjournals.molbev.a026160
Louwhoff SHJJ, Crisp MD (2000) Phylogenetic Analysis of Parmotrema (Parmeliaceae: Lichenized Ascomycotina). Bryologist 103:541–554. https://doi.org/10.1639/0007-2745(2000)103[0541:PAOPPL]2.0.CO;2
Lücking R (2019) Stop the abuse of time! strict temporal banding is not the future of rank-based classifications in fungi (including lichens) and other organisms. Crit Rev Pl Sci 38:199–253. https://doi.org/10.1080/07352689.2019.1650517
Lücking R, Hodkinson BP, Leavitt SD (2017) The 2016 classification of lichenized fungi in the Ascomycota and Basidiomycota – approaching one thousand genera. Bryologist 119:361–416. https://doi.org/10.1639/0007-2745-119.4.361
Mark K, Saag L, Saag A, Thell A, Randlane T (2012) Testing morphology-based delimitation of Vulpicida juniperinus and V. tubulosus (Parmeliaceae) using three molecular markers. Lichenologist 44:757–772. https://doi.org/10.1017/S0024282912000448
Mark K, Randlane T, Thor G, Hur J-S, Obermayer W, Saag A (2019) Lichen chemistry is concordant with multilocus gene genealogy in the genus Cetrelia (Parmeliaceae, Ascomycota). Fungal Biol 123:125–139. https://doi.org/10.1016/j.funbio.2018.11.013
Mattsson J-E, Lai M-J (1993) Vulpicida, a new genus in Parmeliaceae (Lichenized Ascomycetes). Mycotaxon 46:425–428
McCune B, Arup U, Breuss O, Di Meglio E, Di Meglio J, Esslinger TL, Magain N, Miadlikowska J, Miller AE, Muggia L et al (2018) Biodiversity and ecology of lichens of Katmai and Lake Clark National Parks and Preserves, Alaska. Mycosphere 9:859–930. https://doi.org/10.5943/mycosphere/9/4/10
Minh BQ, Nguyen MAT, von Haeseler A (2013) Ultrafast approximation for phylogenetic bootstrap. Molec Biol Evol 30:1188–1195. https://doi.org/10.1093/molbev/mst024
Nelsen MP, Chavez N, Sackett-Hermann E, Thell A, Randlane T, Divakar PK, Rico VJ, Lumbsch HT (2011) The cetrarioid core group revisited (Lecanorales: Parmeliaceae). Lichenologist 43:537–551. https://doi.org/10.1017/S0024282911000508
Nguyen L-T, Schmidt HA, von Haeseler A, Minh BQ (2015) IQ-TREE: a fast and effective stochastic algorithm for estimating maximum likelihood phylogenies. Molec Biol Evol 32:268–274. https://doi.org/10.1093/molbev/msu300
Prokopiev IA, Poryadina LN, Konoreva LA, Chesnokov SV, Shavarda AL (2018) Variation in the composition of secondary metabolites in Flavocetraria lichens from Eastern Siberia. Russ J Ecol 49:401–405. https://doi.org/10.1134/S1067413618050107
Rico VJ, van den Boom PPG, Barrasa JM (2005) Morphology, chemistry and distribution of Melanelia sorediella (Parmeliaceae) and similar species in the Iberian Peninsula. Lichenologist 37:199–215. https://doi.org/10.1017/S0024282905014830
Rodrigues AS, Canêz LS, Lorenz AP (2021) Canoparmelia amazonica, Myelochroa lindmanii and Parmelinella salacinifera belong to Parmelinella (Parmeliaceae). Bryologist 124:352–361. https://doi.org/10.1639/0007-2745-124.3.352
Ronquist R, Teslenko M, van der Mark P, Ayres DL, Darling A, Höhna S, Larget B, Liu L, Suchard MA, Huelsenbeck JP (2012) MrBayes 3.2: efficient bayesian phylogenetic inference and model choice across a large model space. Syst Biol 61:539–542. https://doi.org/10.1093/sysbio/sys029
Saag A, Randlane T, Saag L, Thell A, Ahti T (2013) Third world list of cetrarioid lichens: a databased tool for documentation of nomenclatural data—lessons learned. Taxon 62:591–603. https://doi.org/10.12705/623.4
Saag L, Tõrra T, Saag A, Del-Prado R, Randlane T (2011) Phylogenetic relations of European shrubby taxa of the genus Usnea. Lichenologist 43:427–444. https://doi.org/10.1017/S0024282911000375
Sela I, Ashkenazy H, Katoh K, Pupko T (2015) GUIDANCE2: accurate detection of unreliable alignment regions accounting for the uncertainty of multiple parameters. Nucl Acids Res 43:W7–W14. https://doi.org/10.1093/nar/gkv318
Smith CW, Aptroot A, Coppins BJ, Fletcher A, Gilbert OL, James PW, Wolseley PA (eds) (2009) The Lichens of Great Britain and Ireland. The British Lichen Society, London
Thell A, Feuerer T, Kärnefelt I, Myllys L, Stenroos S (2002a) Phylogeny and ecology of Cetraria obtusata, Coelopogon epiphorellus, and related taxa (Parmeliaceae, lichenized ascomycetes). Mitt Inst Allg Bot Hamburg 30–32:283–296
Thell A, Crespo A, Divakar PK, Kärnefelt I, Leavitt SD, Lumbsch HT, Seaward MRD (2012) A review of the lichen family Parmeliaceae – history, phylogeny and current taxonomy. Nordic J Bot 30:641–664. https://doi.org/10.1111/j.1756-1051.2012.00008.x
Thell A, Feuerer T, Kärnefelt I, Myllys L, Stenroos S (2004) Monophyletic groups within the Parmeliaceae identified by ITS rDNA, β-tubulin and GAPDH sequences. Mycol Progr 3:297–314. https://doi.org/10.1007/s11557-006-0100-1
Thell A, Stenroos S, Feuerer T, Kärnefelt I, Myllys L, Hyvönen J (2002b) Phylogeny of cetrarioid lichens (Parmeliaceae) inferred from ITS and β-tubulin sequences, morphology, anatomy and secondary chemistry. Mycol Progr 1:335–354. https://doi.org/10.1007/s11557-006-0031-x
Thell A, Högnabba F, Elix JA, Feuerer T, Kärnefelt I, Myllys L, Randlane T, Saag A, Stenroos S, Ahti T, Seaward MRD (2009) Phylogeny of the cetrarioid core (Parmeliaceae) based on five genetic markers. Lichenologist 41:489–511. https://doi.org/10.1017/S0024282909990090
Thell A, Kärnefelt I, Seaward MRD (2018) Splitting or synonymizing – genus concept and taxonomy exemplified by the Parmeliaceae in the Nordic region. Graphis Scripta 30:130–137
Trifinopoulos J, Nguyen L-T, von Haeseler A, Minh BQ (2016) W-IQ-TREE: a fast online phylogenetic tool for maximum likelihood analysis. Nucl Acids Res 44(W1):W232–W235. https://doi.org/10.1093/nar/gkw256
van den Boom PPG, Sipman HJM (1994) Cetraria obtusata comb. et stat. nov., an overlooked lichen species from the Central Alps. Lichenologist 26:105–112. https://doi.org/10.1006/lich.1994.1010
White TJ, Bruns T, Lee S, Taylor JW (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR protocols: a guide to methods and applications. Academic Press, New York, pp 315–322. https://doi.org/10.1016/B978-0-12-372180-8.50042-1
Zheludeva EV (2017) New records of lichen species from Magadan region. Turczaninowia 20:64–74. https://doi.org/10.14258/turczaninowia.20.2.6 (In Russian)
Zheludeva EV, Makryi TV (2016) Originality of the lichen flora of Northern Okhotomorye (Region of the northern coast of the Sea of Okhotsk): specific and rare species. In: Shmakov AI, Kopytina TM (eds) Proceedings of the 15th international scientific and practical conference (Barnaul, 23–26 May 2016). Concept, Barnaul, pp 373–380 (In Russian)
Zhurbenko MP, Laursen GA, Walker DA (2005) New and rare lichenicolous fungi and lichens from the North American Arctic. Mycotaxon 92:201–212
Zoller S, Scheidegger C, Sperisen C (1999) PCR primers for the amplification of mitochondrial small subunit ribosomal DNA of lichen-forming ascomycetes. Lichenologist 31:511–516. https://doi.org/10.1006/lich.1999.0220
Acknowledgements
Thanks to G.S. Evdokimov, I.S. Stepanchikova (St. Petersburg State University, St. Petersburg, Russia) and A.P. Yatsyna (V.F. Kuprevich Institute of Experimental Botany NAS B, Minsk, Belarus) for the fresh specimens of Flavocetraria cucullata and Foveolaria nivalis. We thank colleagues from Finland for the opportunity to work with the lichen collections in 2018–2019. We are also grateful to Prof. Mark Seaward (University of Bradford, United Kingdom) for the linguistic revision of the manuscript and anonymous reviewers for valuable comments.
Funding
The study was partly supported by the Russian Foundation for Basic Research, RFBR (Grant No. 18-34-00332, S. Chesnokov, I. Prokopiev; Grant No. 18-05-60093, S. Chesnokov, L. Konoreva). The study was carried out in the frame of institutional research projects “Cryptogamic biota of Pacific Asia: taxonomy, biodiversity, species distribution” of the Botanical Garden-Institute of Far Eastern Branch of the Russian Academy of Sciences (work by S. Chesnokov, L. Konoreva); “Flora and taxonomy of algae, lichens and bryophytes of Russia and phytogeographically important regions of the world” No. 121021600184-6 (work by S. Chesnokov) and “Assessment of changes in the correlation structure of metabolite networks in the process of growth and development of fungi and plants from the standpoint of system biology” No. AAAA-A18-118032390136-5 (work by A. Shavarda, I. Prokopiev) of the Komarov Botanical Institute of the Russian Academy of Sciences; “Flora of lichens, cyanoprokaryotes, bryophytes and vascular plants of the European Arctic and Subarctic” No. AAAA-A18-118050490088-0 (work by L. Konoreva) of the Avrorin Polar-Alpine Botanical Garden-Institute of the Russian Academy of Sciences; “Physiological and biochemical mechanisms of adaptation of plants, animals, humans to the conditions of the Arctic/Subarctic and the development of biological products based on natural northern raw materials that increase the efficiency of the adaptation process and the level of human health in extreme environmental conditions” No. AAAA-A21-121012190035-9 (work by I. Prokopiev) and “Vegetation cover of the permafrost zone of the taiga Yakutia: biodiversity, habitat-forming functions, protection and rational use” No. AAAA-A21-121012190038-0 (work by L. Poryadina) of the Institute for Biological Problems of the Cryolithozone, Siberian Branch, Russian Academy of Sciences with the use of scientific equipment of the Shared core facilities of the Federal Research Center «Yakutsk Science Center SB RAS» within the framework of the implementation of activities under grant No. 13.21.0016; “Inventory and classification of the taxonomic spatial diversity of plants and plant communities in the North of the Russian Far East” No. AAA-A17-117122590002-0 (work by E. Zheludeva) of the Institute of biological problems of the North of Far Eastern Branch of the Russian Academy of Sciences.
Author information
Authors and Affiliations
Contributions
Sergey V. Chesnokov and Liudmila A. Konoreva wrote the main manuscript text, prepared figures 1-5 and table 4; Evgeny A. Davydov performed phylogenetic analysis using IQ-TREE and prepared tables 1 and 2; Ilya A. Prokopiev and Alexey L. Shavarda performed the liquid chromatography–mass spectrometry (LC-MS) metabolite profiling and prepared table 3; Lena N. Poryadina and Elena V. Zheludeva conducted a morphological analysis of specimens in herbaria MAG, SASY and took part in editing the text of the manuscript.
Corresponding author
Additional information
Handling Editor: Miroslav Kolarik.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chesnokov, S.V., Davydov, E.A., Konoreva, L.A. et al. The monotypic genus Flavocetraria and two new genera: Cladocetraria and Foveolaria, in the cetrarioid core. Plant Syst Evol 309, 24 (2023). https://doi.org/10.1007/s00606-023-01862-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00606-023-01862-2