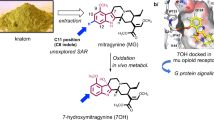
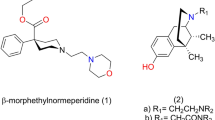
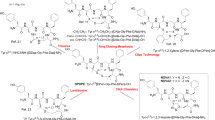
Abstract
Among analgesic drugs, the opioid class of compounds still remains one of the most important medicines for severe and chronic pain treatment. Hence, developing novel and effective synthetic method of morphine and its related compounds is still an important task in modern synthetic organic chemistry. Achieving this goal demands a comprehensive knowledge of these valuable alkaloids. The present review study aims to summarize the history of five major opioid alkaloids and their pharmacologic effects, as well as various synthetic and biosynthetic methods.












































Similar content being viewed by others
References
P.R. Blakemore, J.D. White, Morphine, the Proteus of organic molecules. Chem. Commun. 11, 1159–1168 (2002)
G.D. Busse, D.J. Triggle, Morphine, Drugs: The Straight Facts, (Chelsea House Publishing, 2006), pp. 8–18
G.M. Heyman, “Opiate Use and Abuse, History of”, International Encyclopedia of the Social & Behavioral Sciences, 2nd edn. (Elsevier, Oxford, 2015), pp. 236–242
P.L. Schiff Jr., Opium and its alkaloids. Am. J. Pharm. Educ. 66, 186–193 (2002)
D. Mani, S.S. Dhawan, Scientific basis of therapeutic uses of opium poppy (PapaverSomniferum) in a Ayurveda. Acta Hort. 1036, 175–180 (2014)
J.P. Hoffmann, The historical shift in the perception of opiates: from medicine to social menace. J. Psychoact. Drugs 22, 53–62 (1990)
J.M. Brownstein, A brief history of opiates, opioid peptides, and opioid receptors. Proc. Natl. Acad. Sci. USA 90, 5391–5393 (1993)
L.W. Fleming, A medical bouquet poppies, cinchona and willow. Scot. Med. J. 44, 176–179 (1999)
M. Obladen, Lethal lullabies: a history of opium usein infants. J. Hum. Lact. 32, 75–85 (2015)
A. Moosavyzadeh, F. Ghaffari, S.H. Mosavat, A. Zargaran, A. Mokri, S. Faghihzadeh, M. Naseri, The medieval Persian manuscript of Afyunieh: the first individual treatise on the opium and addiction in history. J. Integr. Med. 16, 77–83 (2018)
J.C. Kramer, Opium rampant: medical use, misuse and abuse in britain and the west in the 17th and 18th centuries. Addiction 74, 377–389 (1979)
G.B. Stefano, N. Pilonis, R. Ptacek, R.M. Kream, Reciprocal evolution of opiate science from medical and cultural perspectives. Med. Sci. Monit. 23, 2890–2896 (2017)
D. Macht, The history of opium and some of its preparations and alkaloids. JAMA 64, 477–481 (1915)
G.W. Pasternak, Y.X. Pan, Mu opioids and their receptors: evolution of a concept. Pharmacol. Rev. 65, 1257–1317 (2013)
K. Brook, J. Bennett, S.P. Desai, The structure of morphine: an 8000-year journey, from resin to de-novo synthesis. J. Anesth. Hist. 3(2), 50–55 (2017)
M. Heydari, H.M. Hashempur, A. Zargaran, Medicinal aspects of opium as described in Avicenna’s Canon of Medicine. Acta Med. Hist. Adriat. 11(1), 101–112 (2013)
F. Ghaffari, M. Naseri, M. Movahhed, A. Zargaran, Spinal traumas and their treatments according to Avicenna’s Canon of Medicine. World Neurosurg. 84(1), 173–177 (2015)
S. Hamarneh, Pharmacy in medieval Islam and the history of drug addiction. Med Hist. 16(3), 226–237 (1972)
H. Khademi, F. Kamangar, P. Brennan, R. Malekzadeh, Opioid therapy and its side effects: a review. Arch. Iran Med. 19, 870–876 (2016)
A. Rosenblum, L.A. Marsch, H. Joseph, R.K. Portenoy, Opioids and the treatment of chronic pain: controversies, current status, and future directions. Exp. Clin. Psychopharmacol. 16, 405 (2008)
J.J. Hobbs, Troubling fields: the opium poppy in Egypt. Geogr. Rev. 88(1), 64–85 (2010)
P.G. Kritikos, S.P. Papadaki, The history of the poppy and of opium and their expansion in antiquity in the eastern Mediterranean area. Bull. Narc. 19(3), 17–38 (1967)
S. Neal, Opium and migration: jardine matheson’s imperial connections and the recruitment of Chinese labour for Assam, 1834–39. Mod. Asian Stud. 51, 1626–1655 (2017)
K. Reist, Opium wars (1839–1842, 1856–1860), in The encyclopedia of war, 1st edn. (Blackwell Publishing Ltd, Hoboken, 2011)
M. Bagheria, M.R. Taheria, M. Farhadpoura, H. Rezadoosta, A. Ghassempoura, H.Y. Aboul-Eneinb, Evaluation of hydrophilic interaction liquid chromatographystationary phases for analysis of opium alkaloids. J. Chromatogr. A 1511, 77–84 (2017)
E. Dehghan, B. Hosseini, H. Naghdi Badi, F. Shahriari Ahmadi, Application of conventional and new biotechnological approaches for improving of morphinane alkaloids production. J. Med. Plant. 9, 34–50 (2010)
A.L. Devereaux, S.L. Mercer, C.W. Cunningham, DARK classics in chemical neuroscience: morphine. ACS Chem. Neurosci. 9, 2395–2407 (2018)
E. Martinez-Fernandez, F. Aragon-Poce, C. Marquez-Espinos, A. Perez-Perez, The history of opiates. Int. Congr. Ser. 1242, 75–77 (2002)
T.A. Alston, Morphine zwitterion. J. Anesth. Hist. 3, 69–70 (2017)
M. Freemantle, Morphine. Chem. Eng. News. 83, 90 (2005)
T. Anderson, On the chemistry of opium. J. Chem. Soc. 15, 446–455 (1862)
Oripavine, 34th ECDD, 6/3 (2006)
P. Madadi, G. Koren, Pharmacogenetic insights into codeine analgesia: implications to pediatric codeine use. Pharmacogenomics 9(9), 1267–1284 (2008)
T.I. Kakhia, Alkaloids & Alkaloids Plants, (Industry Joint Research Center-Adana University-Turkey, 2012), pp. 295–338
J.M.P.J. Garrido, C. Delerue-Matos, F. Borges, T.R.A. Macedo, A.M. Oliveira-Brett, Voltammetric oxidation of drugs of abuse, (II) codeine and metabolites. Electroanalysis 16(17), 1427–1433 (2004)
N. MacDonald, S.M. MacLeod, Has the time come to phase out codeine? CMAJ 182(17), 1825–1825 (2010)
C. Straube, S. Derry, K.C. Jackson, P.J. Wiffen, R.F. Bell, S. Strassels, S. Straube, Codeine, alone and with paracetamol (acetaminophen), for cancer pain (review). Cochrane Database Sys. Rev. 9, CD006601 (2014)
M.L. Fleming, M.A. Wanat, To prescribe codeine or not to prescribe codeine? J. Pain Palliat. Care Pharmacother. 28(3), 251–254 (2014)
S.C. Armstrong, K.L. Cozza, Pharmacokinetic drug interactions of morphine, codeine, and their derivatives: theory and clinical reality, part II. Psychosomatics 44(6), 515–520 (2003)
Q.Y. Yue, J.O. Svensson, C. Alm, F. Sjöqvist, J. Säwe, Interindividual and interethnic differences in the demethylation and glucuronidation of codeine. Br. J. Clin. Pharm. 28, 629–637 (1989)
R.B. Barber, H. Rapoport, Synthesis of thebaine and oripavine from codeine and morphine. J. Med. Chem. 18, 1074–1077 (1975)
R.E. Lister, Structure-activity requirements in some novel Thebaine-derived analgesics. J. Pharm. Pharmacol. 16(5), 364–366 (1964)
A. Lipp, D. Ferenc, C. Gütz, M. Geffe, N. Vierengel, D. Schollmeyer, H.J. Schäfer, S.R. Waldvogel, T. Opatz, A regio- and diastereoselective anodic Aryl-Aryl coupling in the biomimetic total synthesis of (−)-thebaine. Angew. Chem. 57(34), 11055–11059 (2018)
S.C. Pinzaru, N. Leopold, I. Pavel, W. Kiefer, Raman, SERS and theoretical studies of papaverine hydrochloride and its neutral species. Spectrochim. Acta A 60, 2021–2028 (2004)
J.C. Abber, T.F. Lue, B.R. Orvis, R.D. McClure, R.D. Williams, Diagnostic tests for impotence: a comparison of papaverine injection with the penile-brachial index and nocturnal penile tumescence monitoring. J. Urol. 135, 923–925 (1986)
X. Han, M. Lamshöft, N. Grobe, X. Ren, A.J. Fist, T.M. Kutchan, M. Spiteller, M.H. Zenk, The biosynthesis of papaverine proceeds via (S)-reticuline. Phytochemistry 71, 1305–1312 (2010)
D.A. Guthrie, Studies on the synthesis of papaverine. Ph.D. Dissertation, McGill University, (1952)
M.R. Gilliss, Papaverine-safety in use. J. Am. Geriatr. Soc. 21, 200–201 (1973)
M.S. Pathy, A.J. Reynolds, Papaverine and hepatotoxicity. Postgrad. Med. J. 56, 488–490 (1980)
J. Yan, J.Q. Mi, J.T. He, Z.Q. Guo, M.P. Zhao, W.B. Chang, Development of an indirect competitive ELISA for the determination of papaverine. Talanta 66, 1005–1011 (2005)
C. Liu, Synthesis of clean cabbage like (111) faceted silver crystals for efficient surface-enhanced raman scattering sensing of papaverine. Anal. Chem. 90(16), 9805–9812 (2018)
P.C.G. Rida, D. LiVecche, A. Ogden, J. Zhou, R. Aneja, The noscapine chronicle: a pharmaco-historic biography of the opiate alkaloid family and its clinical applications. Med. Res. Rev. 35(5), 1072–1096 (2005)
K.P. Nayak, E. Brochmann-Hanssen, E.L. Way, Biological disposition of noscapine I. J. Pharm. Sci. 54, 191–194 (1963)
X. Chen, T.-T.T. Dang, P.J. Facchini, Noscapine comes of age. Phytochemistry 111, 7–13 (2015)
M. Mahmoudian, P. Rahimi-Moghaddam, The anti-cancer activity of noscapine: a review. Recent Pat. Anticancer Drug Discov. 4(1), 92–97 (2009)
Z.R. Yang, M. Liu, X.L. Peng, X.F. Lei, J.X. Zhang, W.G. Dong, Noscapine induces mitochondria-mediated apoptosis in human colon cancer cells in vivo and in vitro. Biochem. Biophys. Res. Commun. 421, 627–633 (2012)
A. Niazi, Adsorptive stripping differential pulse voltammetry for determination of trace amounts of noscapine in human plasma. J. Chin. Chem. Soc. 54, 1195–1200 (2007)
H. Schmidhammer, E. Scherb-Bukowiecki, T.A. Mayer, Synthesis of benzazepine analogues of noscapine. Helv. Chim. Acta 77, 1590–1594 (1994)
A.K. Manchukonda, P.K. Naik, B. Sridhar, S. Kantevari, Synthesis and biological evaluation of novel biaryl type a-noscapine congeners. Bioorg. Med. Chem. Lett. 24, 5752–5757 (2014)
Z.Z. Fang, K.W. Krausz, F. Li, J. Cheng, N. Tanaka, F.J. Gonzalez, Metabolic map and bioactivation of the anti-tumour drug noscapine. Br. J. Pharmacol. 167, 1271–1286 (2012)
S. Klemenc, Noscapine as an adulterant in illicit heroin samples. Forensic Sci. Int. 108, 45–49 (2000)
S. Hosztafi, The history of heroin. Acta Pharm. Hung. 71(2), 233–242 (2001)
J. Merry, A social history of heroin addiction. Br. J. Addict. 70, 307–310 (1975)
J. Strang, T. Groshkova, N. Metrebian, New heroin-assisted treatment - recent evidence and current practices of supervised injectable heroin treatment in Europe and beyond, vol. 11 (European Monitoring Centre for Drugs and Drug Addiction, 2012)
M.G. Miller, N. McCarthy, C.A. O’Boyle, M. Kearney, Continuous subcutaneous infusion of morphine vs. hydromorphone: a controlled trial. J. Pain Symptom Manag. 18, 9–16 (1999)
M.G. Kumar, S. Lin, Hydromorphone: analytical methodologies for its determination. Curr. Anal. Chem. 4, 111–126 (2008)
Y.J. Bao, W. Hou, X.Y. Kong, L. Yang, J. Xia, B.J. Hua, R. Knaggs, Hydromorphone for cancer pain. Cochrane Database Syst. Rev. 10(10), CD011108 (2016)
S.V. Weinstein, A new extended release formulation (OROS®) of hydromorphone in the management of pain. Ther Clin Risk Manag. 5, 75–80 (2009)
E. Prommer, Oxymorphone: a review. Support Care Cancer 14, 109–115 (2006)
F. Mayyas, P. Fayers, S. Kaasa, O. Dale, A systematic review of oxymorphone in the management of chronic pain. J. Pain Symptom Manag. 39, 296–308 (2010)
R.S. Sinatra, D.M. Harrison, A comparison of oxycodone and fentanyl as narcotic supplements in general anesthesia. J. Clin. Anesth. 1, 253–258 (1989)
K.W. Chamberlin, R. Neville, J. Tan, Oral oxymorphone for pain management. Ann. Pharmacother. 41, 1144–1152 (2007)
N. Vadivelu, D. Chang, E.M. Helander, G.J. Bordelon, A. Kai, A.D. Kaye, D. Hsu, D. Bang, I. Julka, Ketorolac, oxymorphone, tapentadol, and tramadol. Anesthesiol. Clin. 35(2), e1–e20 (2017)
X. Ruan, K.F. Mancuso, A.D. Kaye, Revisiting oxycodone analgesia. Anesthesiol. Clin. 35(2), e163–e174 (2017)
K.L. Boyle, C.D. Rosenbaum, Oxycodone overdose in the pediatric population: case files of the university of massachusetts medical toxicology fellowship. J. Med. Toxicol. 10(3), 280–285 (2014)
J.W. Mandema, R.F. Kaiko, B. Oshlack, R.F. Reder, D.R. Stanski, Characterization and validation of a pharmacokinetic model for controlled-release oxycodone. Br. J. Clin. Pharmacol. 42, 747–756 (1996)
H. Kokki, M. Kokki, Central nervous system penetration of the opioid oxycodone, in Neuropathology of Drug Addictions and Substance Misuse. (Elsevier, Amsterdam, 2016), pp. 457–466
E. Kalso, Oxycodone. J. Pain Symptom Manag. 29(5), 47–56 (2005)
R. Huddart, M. Clarke, R.B. Altman, T.E. Klein, Oxycodone pathway, pharmacokinetics. PharmGKB Summary 28(10), 230–237 (2010)
C. Mannich, H. Löwenheim, Ueber zwei neue Reduktionsprodukte des Kodeins. Arch. Pharm. 258, 295–316 (1920)
D. Krashin, N. Murinova, A. Trescot, Extended-release hydrocodone – gift or curse? J. Pain Res. 6, 53–57 (2013)
S.L. Walsh, P.A. Nuzzo, M.R. Lofwall, J.R. Holtman, The relative abuse liability of oral oxycodone, hydrocodone and hydromorphone assessed in prescription opioid abusers. Drug Alcohol Depend. 98, 191–202 (2008)
A. Saitman, R.L. Fitzgerald, L.M. McIntyre, Evaluation and comparison of postmortem hydrocodone concentrations in peripheral blood, central blood and liver specimens: a minimal potential for redistribution. Forensic Sci. Int. 247, 36–40 (2015)
F.S. Alshehri, A.Y. Hakami, Y.S. Althobaiti, Y. Sari, Effects of ceftriaxone on hydrocodone seeking behavior and glial glutamate transporters in rats. Behav. Brain Res. 347, 368–376 (2018)
L. Day, K. Kleinschmidt, M.B. Forrester, S.Y. Feng, Comparison of unintentional exposures to codeine and hydrocodone reported to texas poison centers. J. Emerg. Med. 50, 744–752 (2016)
R.J. Defalque, A.J. Wright, The early history of methadone, myths and facts. Bull. Anesth. Hist. 25(3), 13–16 (2007)
F. Leri, A. Tremblay, R.E. Sorge, J. Stewart, Methadone maintenance reduces heroin- and cocaine-induced relapse without affecting stress-induced relapse in a rodent model of poly-drug use. Neuropsychopharmacology 29, 1312–1320 (2004)
“Methadone: what's the story?” Dublin, UISCE, 32, 1–34 (2003)
A.B.M. Paul, L. Simms, A.M. Mahesan, The toxicology of methadone-related death in infants under 1 year: three case series and review of the literature. J. Forensic Sci. 62, 1414–1417 (2017)
J.M. Corkery, F. Schifano, A.H. Ghodse, A. Oyefeso, The effects of methadone and its role in fatalities. Hum. Psychopharmacol. Clin. Exp. 19(8), 565–576 (2004)
R. Ojha, S.C. Bhatia, Tramadol dependence in a patient with no previous substance history. Prim. Care Companion J. Clin. Psychiatry (2010). https://doi.org/10.4088/PCC.09l00779ecr
Tramadol, Expert Committee on Drug Dependence, Thirty-sixth Meeting. Geneva, WHO Technical Report Series 991 (2014)
V. Varghese, T. Hudicky, A short history of the discovery and development of naltrexone and other morphine derivatives, in Natural Products in Medicinal Chemistry. (Wiley-VCH Verleg GmbH & Co. KGaA, New Jersey, 2014), pp. 225–250
V. Beltran-Campos, M. Silva-Vera, M.L. Garcia-Campos, S. Diaz-Cintra, Effects of morphine on brain plasticity. Neurologia (English Edition) 30(3), 176–180 (2015)
S. Benyhe, F. Zador, F. Otvos, Biochemistry of opioid (morphine) receptors: binding, structure and molecular modeling. Acta Biol Szeged. 59, 17–37 (2015)
P.A.J. Janssen, A review of the chemical features associated with strong morphine-like activity. Br. J. Anaesth. 34, 260–268 (1962)
A.M. Trescot, S. Datta, M. Lee, H. Hansen, Opioid pharmacology. Pain Physician 11, 133–153 (2008)
B.L. Kieffer, C.J. Evans, Opioid receptors: from binding sites to visible molecules in vivo. Neuropharmacology 56, 205–212 (2009)
P.K. Janicki, Pharmacology of Morphine Metabolites. Curr. Pain Headache Rep. 1(264–270), 238 (1997)
A. Laux-Biehlmann, J. Mouheiche, J. Veriepe, Y. Goumon, Endogenous morphine and its metabolites in mammals: history, synthesis, localization and perspectives. Neuroscience 233, 95–117 (2013)
Y. Goumon, A. Laux-Biehlmann, A. Muller, D. Aunis, Central and peripheral endogenous morphine. An R Acad. Nac Farm. 75, 389–418 (2009)
A. Koneru, S. Satyanarayana, S. Rizwan, Current knowledge and future trends of endogenous opioids: three physiological role and receptors. Pharmacologyonline 1, 780–788 (2010)
I. Sora, G. Elmer, M. Funada, J. Pieper, X.F. Li, F.S. Hall, G.R. Uhl, μ opiate receptor gene dose effects on different morphine actions evidence for differential in vivo μ receptor reserve. Neuropsychopharmacology 25(1), 41–54 (2011)
T.P. Su, T.C. Su, Y. Nakamura, S.Y. Tsai, The sigma-1 receptor as a pluripotent modulator in living systems. Trends Pharmacol. Sci. 37(4), 262–278 (2016)
C.G. Rousseaux, S.F. Greene, Sigma receptors [σRs]: biology in normal and diseased states. J. Recept. Signal. Transduct. Res. 36(4), 327–388 (2016)
I.S. Zagon, S.R. Goodman, P.J. McLaughlin, Zeta (ζ), the opioid growth factor receptor: identification and characterization of binding subunits. Brain Res. 605(1), 50–56 (1993)
T. Toubia, T. Khalife, The endogenous opioid system: role and dysfunction caused by opioid therapy. Clin. Obstet. Gynecol. 62, 3–10 (2019)
A. Koneru, S. Satyanarayana, S. Rizwan, Endogenous opioids: their physiological role and receptors. Global J. Pharmacol. 3(3), 149–153 (2009)
T.A. Koshimizu, K. Honda, S. Nagaoka-Uozumi, A. Ichimura, I. Kimura, M. Nakaya, N. Sakai, K. Shibata, K. Ushijima, A. Fujimura, A. Hirasawa, H. Kurose, G. Tsujimoto, A. Tanoue, Y. Takano, Complex formation between the vasopressin 1b receptor, β-arrestin-2, and the μ-opioid receptor underlies morphine tolerance. Nat. Neurosci. 21(6), 820–833 (2018)
H. Pathan, J. Williams, Basic opioid pharmacology: an update. Br. J. Pain 6(1), 11–16 (2012)
D.S. Goodsell, The molecular perspective: morphine. Stem Cells 23(1), 144–145 (2005)
L.A. Chahl, Opioids - mechanisms of action. Exp Clin Pharmacol 19, 63–65 (1996)
L.L. Christrup, Morphine metabolites. Acta Anaesthcsiol. Scand. 41, 116–122 (1997)
G. Andersen, L. Christrup, P. Sjøgren, Relationships among morphine metabolism, pain and side effects during long-term treatment. J. Pain Symptom Manag. 25(1), 74–91 (2003)
J. Lötsch, Opioid metabolites. J. Pain Symptom Manag. 29, S10-24 (2005)
N. Sunagane, T. Ogawa, T. Uruno, K. Kubota, Mechanism of relaxant action of papaverine VI. Sodium ion dependence of its effect on 45Ca-Efflux in Guinea-Pig Taenia Coli. Jpn. J. Pharmacol. 38, 133–139 (1985)
G. Sanfilippo, Contributo sperimentale all'ipotesi della smetilazione della codeine nell'organismo. I . Influenze della dose sull'assuefazione alla codeina. II. Assuefazione alla codeine attenutacon somministrazione prolungata di morphina. Boll. Soc. Ital. Biol. Sper. 24, 723–726 (1948)
D.A. Yee, R.S. Atayee, B.M. Best, J.D. Ma, Observations on the urine metabolic profile of codeine in pain patients. J. Anal. Toxicol. 38(2), 86–91 (2014)
C.H. Mahler, E.D. Stevens, M.L. Trudell, S.P. Nolan, (-)-Thebaine. Acta Cryst. C52, 3193–3195 (1996)
T.T. Tok, S.J.T. Gowder, Structural and pharmacological properties of alkaloids with special reference to thebaine type alkaloids. Biomed. J. Sci. Tech. Res. 17(3), 12767–12780 (2019)
L. Maat, J.A. Peters, M.A. Prazeres, Diels-alder reaction of thebaine via N-formylnorthebaine with nitroethene; reduction of the nitro group in 7α-nitroethenoisomorphinans (chemistry of opium alkaloids, part XX). Recl. Trav. Chim. Pays-Bas. 104(7–8), 205–208 (2010)
H. Rapoport, P. Sheldrick, The diels-alder reaction with thebaine. thermal rearrangement of some adducts from acetylenic dienophiles. J. Am. Chem. Soc. 85(11), 1636–1642 (1963)
H.K. Knutsen, J. Alexander, L. Barregard, M. Bignami, B. Bruschweiler, S. Ceccatelli, B. Cottrill, M. Dinovi, L. Edler, B. Grasl-Kraupp, C. Hogstrand, L. Hoogenboom, C.S. Nebbia, I.P. Oswald, A. Petersen, M. Rose, A.-C. Roudot, T. Schwerdtle, G. Vollmer, H. Wallace, D. Benford, G. Calo, A. Dahan, B. Dusemund, P. Mulder, E. Nemeth-Zamborine, D. Arcella, K. Baert, C. Cascio, S. Levorato, M. Schutte, C. Vleminckx, Update of the scientific opinion on opium alkaloids in poppy seeds. EFSA J. 16(5), 5243–5362 (2018)
M.D. Aceto, L.S. Harris, M.E. Abood, K.C. Rice, Stereoselective μ- and δ-opioid receptor-related antinociception and binding with (+)-thebaine. Eur. J. Pharmacol. 365(2–3), 143–147 (1999)
G. Mikus, A.A. Somogyi, F. Bochner, M. Eichelbaum, ThebaineO-demethylation to oripavine: genetic differences between two rat strains. Xenobiotica 21(11), 1501–1509 (1991)
M.P. Gómez-Serranillos, O.M. Palomino, E. Carretero, A. Villar, Analytical study and analgesic activity of oripavine from Papaver somniferum L. Phytother. Res. 12(5), 346–349 (1998)
B.M. El-Haj, H.S. Ali, N.M. Hamoudi, Oripavine as a new marker of opiate product use. Forensic Toxicol. 29(2), 152–158 (2011)
C.C. Hodges, J.S. Horn, H. Rapoport, Morphinan alkaloids in Papaver bracteatum: Biosynthesis and Fate. Technical Information Division, Lawrence Berkeley National Laboratory, Berkeley No. 94720 (1977)
H. Kodaira, S. Spector, Transformation of thebaine to oripavine, codeine, and morphine by rat liver, kidney, and brain microsomes. Proc. Natl. Acad. Sci. USA 85(4), 1267–1271 (1988)
H. Uprety, D.S. Bhakuni, R.S. Kapil, Biosynthesis of Papaverine. Phytochemistry 14, 1535–1537 (1975)
A.V. Lakyanov, V.S. Onoprienko, V.A. Zasosov, Industrial Methods of Preparing Papaverine (A Review of the Literature) (Chemical and Pharmaceutical Scientific-Research Institute, Moscow, 1972).
S.K. Talapatra, B. Talapatra, Chemistry of Plant Natural Products: Stereochemistry, Conformation, Synthesis, Biology, and Medicine (Spring, Heidelberg New York Dordrecht London, 2015).
C. Hanna, J. Shutt, Papaverine analogs. VI relationship between chemical structure and coronary vasodilator action. Arch. Exp. Pathol. U. Pharmakol. 220, S43-51 (1953)
A. Capasso, S. Piacente, N. Tommasi, L. Rastrelli, C. Pizza, The effect of isoquinoline alkaloids on opiate withdrawal. Curr. Med. Chem. 13(7), 807–812 (2006)
L. Hertle, H. Nawrath, Effects of papaverine on human isolated bladder muscle. Urol. Res. 18, 227–231 (1990)
N. Sunagane, T. Uruno, K. Kubota, Mechanism of relaxant action of papaverine, effect on caffeine-induced contraction of guinea pig taenia coli. Jpn. J. Pharmacol. 32(5), 785–793 (1982)
J.P. Rosazza, M. Kammer, L. Youel, Microbial models of mammalian metabolism O-demethylations of papaverine. Xenobiotica 7, 133–143 (1977)
J. Axelrod, J. Cochin, The inhibitory action of nalorphine on the enzymatic N-demethylation of narcotic drugs. J. Pharmacol. Exp. Ther. 121(1), 107–112 (1957)
F.M. Belpaire, M.G. Bogaert, M.T. Rosseel, Metabolism of papaverine I. Identification of metabolites in rat bile. Xenobiotica 5, 413–420 (1975)
F.M. Belpaire, M.T. Rosseel, M.G. Bogaert, Metabolism of papaverine IV. Urinary elimination of papaverine metabolites in man. Xenobiotica 8, 297–300 (1978)
G. Wilen, P. Ylitalo, Metabolism of [14C] papaverine in man. J. Pharm. Pharmacol. 34, 264–266 (1982)
J.C. Davila, C.G. Reddy, P.J. Davis, D. Acosta, Toxicity assessment of paraverine hydrochloride and papaverine-derived metabolites in primary cultures of rat hepatocytes. In Vitro Cell Dev. Biol. 26(5), 515–524 (1990)
M.A. Al-Yahya, M.M.A. Hassan, Noscapine. Anal. Profiles Drug Subst. 11, 407–461 (1982)
S. Cheriyamundath, T. Mahaddalkar, P.K. Reddy Nagireddy, B. Sridhar, S. Kantevari, M. Lopus, Insights into the structure and tubulin-targeted anticancer potential of N-(3-bromobenzyl) noscapine. Pharmacol. Rep. 1, 48–53 (2019)
M. Tripathi, P.L. Reddy, D.S. Rawat, Noscapine and its analogues as anti-cancer agents. Chem. Biol. Interface 4, 1–22 (2014)
N. Dhiman, A. Sood, A. Sharma, Noscapine: an anti-mitotic agent. World J. Pharm. Pharm. Sci. 3, 324–338 (2013)
K. Ye, Y. Ke, N. Keshava, J. Shanks, J.A. Kapp, R.R. Tekmal, J. Petros, H.C. Joshi, Opium alkaloid noscapine is an antitumor agent that arrests metaphase and induces apoptosis in dividing cells. Proc. Natl. Acad. Sci. USA 95, 1601–1606 (1998)
L. Richards, A. Lutz, D.K. Chalmers, A. Jarrold, T. Bowser, G.W. Stevens, S.L. Gras, Production of metabolites of the anti-cancer drug noscapine using a P450BM3 mutant library. Biotechnol. Rep. 24, e00372 (2019)
N. Tsunoda, Metabolic fate of noscapine.11.Isolation and identification of novel metabolites produce4 by C-C bond cleavage. Xenobiotica 9, 181–187 (1979)
A.O. Alnajjar, M.E. El-Zaria, Synthesis and characterization of novel azo-morphine derivatives for possible use in abused drugs analysis. Eur. J. Med. Chem. 43, 357–363 (2008)
D.A. Rincón, M.N.D.S. Cordeiro, R.A. Mosquera, Theoretical study of morphine and heroin: conformational study in gas phase and aqueous solution and electron distribution analysis. Int. J. Quantum Chem. 110, 2472–2482 (2010)
J.J. Rady, G.I. Elmer, G.M. Fujimoto, Opioid receptor selectivity of heroin given intracerebroventricularly differs in six strains of inbred mice. J. Pharmacol. Exp. Ther. 288, 438–445 (1999)
J.J. Rady, F. Aksu, J.M. Fujimoto, The heroin metabolite, 6-monoacetylmorphine, activates delta opioid receptors to produce antinociception in Swiss-Webster mice. J. Pharmacol. Exp. Ther. 268(3), 1222–1231 (1994)
D.E. Selley, C.C. Cao, T. Sexton, J.A. Schwegel, T.J. Martin, S.R. Childers, mu Opioid receptor-mediated G-protein activation by heroin metabolites: evidence for greater efficacy of 6-monoacetylmorphine compared with morphine. Biochem. Pharmacol. 62(4), 447–455 (2001)
H.H. Maurer, C. Sauer, D.S. Theobald, Toxicokinetics of drugs of abuse: current knowledge of the isoenzymes involved in the human metabolism of tetrahydrocannabinol, cocaine, heroin, morphine, and codeine. Lippincott Williams Wilkins 28, 447–453 (2006)
A. Murray, N.A. Hagen, Hydromorphone. J. Pain Symptom Manag. 29, S57–S66 (2005)
N.H. Barakat, R.S. Atayee, M.B. Best, J.D. Ma, Urinary hydrocodone and metabolite distributions in pain patients. J. Anal. Toxicol. 38, 404–409 (2014)
S. Valtier, V.S. Bebarta, Excretion profile of hydrocodone, hydromorphone and norhydrocodone in urine following single dose administration of hydrocodone to healthy volunteers. J. Anal. Toxicol. 36, 507–514 (2012)
P. Gulur, K. Koury, P. Arnstein, H. Lee, P. McCarthy, C. Coley, E. Mort, Morphine versus hydromorphone: does choice of opioid influence outcomes? Pain Res. Treat. 2015, 1–6 (2015)
B. Lalovic, E. Kharasch, C. Hoffer, L. Risler, L. Liuchen, D. Shen, Pharmacokinetics and pharmacodynamics of oral oxycodone in healthy human subjects: role of circulating active metabolites. Clin. Pharmacol. Ther. 79(5), 461–479 (2006)
M. Kokki, M. Heikkinen, P. Välitalo, H. Hautajärvi, J. Hokkanen, H. Pitkänen, U. Sankilampi, V.-P. Ranta, H. Kokki, Maturation of oxycodone pharmacokinetics in neonates and infants: oxycodone and its metabolites in plasma and urine. Br. J. Clin. Pharm. 83(4), 791–800 (2016)
A.Z. DePriest, R. Heltsley, D.L. Black, J.M. Mitchell, C. LoDico, R. Flegel, E.J. Cone, Prescription opioids. V. Metabolism and excretion of oxymorphone in urine following controlled single dose administration. J. Anal. Toxicol. 40(8), 566–574 (2016)
H.S. Smith, Clinical pharmacology of oxymorphone. Pain Med. 10, S3–S10 (2009)
D.S. Craig, Oxymorphone extended-release tablets (Opana ER) for the management of chronic pain. P T 35(6), 324–329 (2010)
S.M. Fishman, B. Wilsey, G. Mahajan, P. Molina, Methadone reincarnated: novel clinical applications with related concerns. Pain Med. 3, 339–348 (2002)
P. Lisberg, F. Scheinmann, Is it time to consider use of levo-methadone (R-(-)-Methadone) to replace racemic methadone? J. Dev. Drugs. 2, 1–2 (2013)
N. Ansermot, Substitution of (R, S)-methadone by (R)-methadone. Arch. Intern. Med. 170(6), 529–536 (2010)
D.A.N. Silverman, R.T. Nettleton, K.B. Spencer, M. Wallisch, G.D. Olsen, S-Methadone augments R-methadone induced respiratory depression in the neonatal guinea pig. Respir. Physiol. Neurobiol. 169(3), 252–261 (2009)
E.F. Mccance-Katz, (R)-methadone versus racemic methadone: what is best for patient care? Addiction 106(4), 687–688 (2011)
J. Gaertner, R. Voltz, C. Ostgathe, Methadone: a closer look at the controversy. J. Pain Symptom Manage. 36(2), e4–e7 (2008)
S. Gadel, C. Friedel, E.D. Kharasch, Differences in methadone metabolism by CYP2B6 variants. Drug Metab. Dispos. 43(7), 994–1001 (2015)
M.E.M. Larson, T.M. Richards, Quantification of a methadone metabolite (EDDP) in urine: assessment of compliance. Clin. Med. Res. 7(4), 134–141 (2009)
K.L. Preston, D.H. Epstein, D. Davoudzadeh, M.A. Huestis, Methadone and metabolite urine concentrations in patients maintained on methadone. J. Anal. Toxicol. 27(6), 332–341 (2003)
Tramadol Update Review Report, 36th ECDD, (2014)
M. Subedi, S. Bajaj, M.S. Kumar, Y.C. Mayur, An overview of tramadol and its usage in pain management and future perspective. Biomed. Pharmacother. 111, 443–451 (2019)
Q. Shen, Y. Qian, X. Xu, W. Li, J. Liu, W. Fu, Design, synthesis and biological evaluation of N-phenylalkyl-substituted tramadol derivatives as novel μ opioid receptor ligands. Acta Pharmacol. Sin. 36(7), 887–894 (2015)
J. Marcotte, Formal Synthesis of (+/-) Morphine via an Oxy-cope/Claisen/Ene Reaction Cascade. PhD Dissertation, The University of Ottawa, (2012)
D.F. Taber, T.D. Neubert, M.F. Schlecht, in Strategies and Tactics in Organic Synthesis, Chap. 11, vol. 5, ed. by M. Harmata (Elsevier, Amsterdam, 2004), pp. 353–389
L.M. Mascavage, M.L. Wilson, D.R. Dalton, Syntheses of morphine and codeine (1992–2002), templates for exploration of synthetic tools. Curr. Org. Synth. 3, 99–120 (2006)
K.A. Parker, D. Fokas, Convergent synthesis of (.+-.)-dihydroisocodeine in 11 steps by the tandem radical cyclization strategy, a formal total synthesis of (.+-.)-morphine. J. Am. Chem. Soc. 114(24), 9688–9689 (1992)
J.D. White, P. Hmciar, F. Stappenbeck, Asymmetric synthesis of (+)-morphine. The phenanthrene route revisited. J. Org. Chem. 62, 5250–5251 (1997)
D. Trauner, J.W. Bats, A. Werner, J. Mulzer, Synthesis of enantiomerically pure morphine alkaloids: the hydrophenanthrene route. J. Org. Chem. 63, 5908–5918 (1998)
Q. Li, H. Zhang, Total synthesis of codeine. Chem-Eur J. 21(46), 16379–16382 (2015)
D.F. Taber, T.D. Neubert, A. Rheingold, Synthesis of (-)-morphine. J. Am. Chem. Soc. 124, 12416–12417 (2002)
A.T. Omori, K.J. Finn, H. Leisch, R.J. Carroll, T. Hudlicky, Chemoenzymatic total synthesis of (+)-codeine by sequential intramolecular Heck cyclizations via C-B–D ring construction. Synlett 18, 2859–2862 (2007)
G. Stork, A. Yamashita, J. Adams, G.R. Schulte, R. Chesworth, Y. Miyazaki, J.J. Farmer, Regiospecific and stereoselective syntheses of (+/-) morphine, codeine, and thebaine via a highly stereocontrolled intramolecular 4 + 2 cycloaddition leading to a phenanthrofuran system. J. Am. Chem. Soc. 131(32), 11402–11406 (2009)
P. Magnus, N. Sane, B.P. Fauber, V. Lynch, Concise syntheses of (−)-galanthamine and (±)-codeine via intramolecular alkylation of a phenol derivative. J. Am. Chem. Soc. 131(44), 16045–16047 (2009)
H. Koizumi, S. Yokoshima, T. Fukuyama, Total synthesis of (−)-morphine. Chem. Asian J. 5(10), 2192–2198 (2010)
M. Tissot, R.J. Phipps, C. Lucas, R.M. Leon, R.D.M. Pace, T. Ngouansavanh, M.J. Gaunt, Gram-scale enantioselective formal synthesis of morphine through anortho-paraoxidative phenolic coupling strategy. Angew. Chem. 53(49), 13498–13501 (2014)
S. Chu, N. Münster, T. Balan, M.D. Smith, A Cascade strategy enables a total synthesis of (±)-morphine. Angew. Chem. 55(46), 14306–14309 (2016)
H. Umihara, S. Yokoshima, M. Inoue, T. Fukuyama, Total synthesis of (−)-morphine. Chem. Eur. J. 23(29), 6993–6995 (2017)
J. Rautschek, A. Jäger, P. Metz, Formal synthesis of (−)-codeine by application of temporary Thio derivatization. Org. Lett. 20(3), 832–835 (2018)
Q. Zhang, F.-M. Zhang, C.-S. Zhang, S.-Z. Liu, J.-M. Tian, S.-H. Wang, X.-M. Zhang, Y.-Q. Tu, Enantioselective synthesis of cis-hydrobenzofurans bearing all-carbon quaternary stereocenters and application to total synthesis of (-)-morphine. Nat. Commun. 10(1), 1–7 (2019)
J. Brousseau, A. Xolin, L. Barriault, A nine-step formal synthesis of (±)-morphine. Org. Lett. 21(5), 1347–1349 (2019)
H. Rapoport, C.H. Lovell, H.R. Reist, M.E. Warren, The synthesis of thebaine and northebaine from codeinone dimethyl ketal. J. Am. Chem. Soc. 89(8), 1942–1947 (1967)
I. Seki, Studies on the morphine alkaloids and its related compounds. XVII. One-step preparations of enol ether and pyrrolidinyl dienamine of normorphinone derivatives. Chem. Pharm. Bull. 18(4), 671–676 (1970)
A. Coop, K.C. Rice, A novel synthesis of thebaine from codeine. Heterocycles 49, 43–47 (1998)
R.D. Singera, P.J. Scammellsb, Alternative methods for the MnO2 oxidation of codeine methylether to thebaine utilizing ionic liquids. Tetrahedron Lett. 42, 6831–6833 (2001)
S. Hosztafi, Recent advances in the chemistry of oripavine and its derivatives. ABB 5, 704–717 (2014)
M. Geffe, T. Opatz, Enantioselective synthesis of (−)-dihydrocodeine and formal synthesis of (−)-thebaine, (−)-codeine, and (−)-morphine from a deprotonated α-aminonitrile. Org. Lett. 16, 5282–5285 (2014)
P. Koukal, J. Hajicek, S. Gupta, T. Hudlicky, Model studies toward the total synthesis of thebaine by an intramolecular cycloaddition strategy. ChemistrySelect 2(26), 7783–7786 (2017)
D.A. Guthrie, A.W. Frank, C.B. Purves, Studies in the polyoxyphenol series IX, the synthesis of papaverine and papaveraldine by the Pomeranz-Fritsch method. Can. J. Chem. 33, 729–742 (1955)
G. Wu, A.L. Rhiengold, S.J. Geib, R.F. Heck, Palladium-catalyzed annulation of aryl iodides with diphenylacetylene. Organometallics 1987, 6 (1941)
K.R. Roesch, R.C. Larock, Synthesis of isoquinolines and pyridines by the palladium/copper-catalyzed coupling and cyclization of terminal acetylenes and unsaturated imines: the total synthesis of decumbenine B. J. Org. Chem. 67(1), 86–94 (2002)
R.P. Korivi, C.-H. Cheng, Highly efficient synthesis of isoquinolines via nickel-catalyzed annulation of 2-iodobenzaldimines with alkynes: evidence for dual pathways of alkyne insertion. Org. Lett. 7(23), 5179–5182 (2005)
S. Zhang, D. Huang, G. Xu, S. Cao, R. Wang, S. Peng, J. Sun, An efficient synthesis of isoquinolines via rhodium-catalyzed direct C-H functionalization of arylhydrazines. Org. Biomol. Chem. 13(29), 7920–7923 (2015)
C.D. Gilmore, K.M. Allan, B.M. Stoltz, Orthogonal synthesis of indolines and isoquinolines via aryne annulation. J. Am. Chem. Soc. 130, 1558–1559 (2008)
M.V. Madhubabu, R. Shankara, R. Akulaa, U.K. Syam Kumar, M.V.B. Rao, Facile synthesis of papaverine, (±) setigeridine, (±) setigerine, and related isoquinoline alkaloids. Der. Pharma. Chem. 6(4), 50–56 (2014)
P.E. Ghaly, R.M. Abou El-Magd, C.D.M. Churchill, J.M. Tuszynski, F.G. West, A new antiproliferative noscapine analogue: chemical synthesis and biological evaluation. Oncotarget 7, 40518–40530 (2016)
D.U. Lee, (-)-β-Narcotine: a facile synthesis and the degradation with ethyl chloroformate. Bull. Korean Chem. Soc. 23(11), 1548–1552 (2002)
M.D.P.C. Soriano, N. Shankaraiah, L.S. Santos, Short synthesis of noscapine, bicuculline, egenine, capnoidine, and corytensine alkaloids through the addition of 1-siloxy-isobenzofurans to imines. Tetrahedron Lett. 51, 1770–1773 (2010)
A.J. Debono, S.J. Mistry, J. Xie, D. Muthiah, J. Phillips, S. Ventura, R. Callaghan, C.W. Pouton, B. Capuano, P.J. Scammells, The synthesis and biological evaluation of multifunctionalised derivatives of noscapine as cytotoxic agents. Chem. Med. Chem. 9, 399–410 (2014)
R. Tomar, A. Sahni, I. Chandra, V. Tomar, R. Chandra, Review of noscapine and its analogues as potential anti-cancer drugs. Mini Rev. Org. Chem. 15, 1–20 (2018)
A.J. Debono, J.H. Xie, S. Ventura, C.W. Pouton, B. Capuano, P.J. Scammells, Synthesis and biological evaluation of N-substituted noscapine analogues. Chem. Med. Chem. 7, 2122–2133 (2012)
K.B. Mishra, R.C. Mishra, V.K. Tiwari, First noscapine glycoconjugates inspired by click chemistry. RSC Adv. 5(64), 51779–51789 (2015)
A.K. Verma, S. Bansal, J. Singh, R.K. Tiwari, V. Kasi Sankar, V. Tandon, R. Chandra, Synthesis and in vitro cytotoxicity of haloderivatives of noscapine. Bioorganic Med. Chem. 14, 6733–6736 (2006)
R. Aneja, S.N. Vangapandu, M. Lopus, V.G. Viswesarappa, N. Dhiman, A. Verma, C. Chandra, D. Panda, H.C. Joshi, Synthesis of microtubule-interfering halogenated noscapine analogs that perturb mitosis in cancer cells followed by cell death. Biochem. Pharmacol. 72, 415–426 (2006)
R.C. Mishra, P. Karna, S.R. Gundala, V. Pannu, R.A. Stanton, K.K. Gupta, M.H. Robinson, M.O. Lopus, L. Wilson, M. Henary, R. Aneja, Second generation benzofuranone ring substituted noscapine analogs: synthesis and biological evaluation. Biochem. Pharmacol. 82, 110–121 (2011)
R.C. Mishra, S.R. Gundala, P. Karna, M. Lopus, K.K. Gupta, M. Nagaraju, D. Hamelberg, V. Tandon, D. Panda, M.D. Reid, R. Aneja, Design, synthesis and biological evaluation of di-substituted noscapine analogs as potent and microtubule-targeted anticancer agents. Bioorg. Med. Chem. Lett. 25(10), 2133–2140 (2015)
S.M. Devine, C. Yong, D. Amenuvegbe, L. Aurelio, D. Muthiah, C.W. Pouton, R. Callaghan, B. Capuano, P.J. Scammells, Synthesis and pharmacological evaluation of noscapine inspired 5-substituted tetrahydroisoquinolines as cytotoxic agents. J. Med. Chem. 61, 8444–8456 (2018)
M. Mohebbi, M. Bararjanian, S.N. Ebrahimi, M. Smieško, P. Salehi, Noscapine derivatives as new chiral catalysts in asymmetric synthesis: highly enantioselective addition of diethylzinc to aldehydes. Synthesis 50, 1841–1848 (2018)
I. Iijima, J. Minamikawa, A.E. Jacobson, A.E. Jacobson, K.C. Rice, Studies in the (+)-morphinan series, 4. A markedly improved synthesis of (+)-morphine. J. Org. Chem. 43(7), 1462–1463 (1978)
C.R.A. Wright, On the action of organic acids and their anhydrides on the natural alkaloids. J. Chem. Soc. 27, 1031–1043 (1847)
L.R. Odell, J. Skopec, A. McCluskey, A ‘cold synthesis’ of heroin and implications in heroin signature analysis Utility of trifluoroacetic/acetic anhydride in the acetylation of morphine. Forensic Sci. Int. 164, 221–229 (2006)
C.C. Clark, A study of procedures for the identification of heroin. J. Forensic Sci. 22, 418–428 (1977)
J. Schwyzer, Die Fabrikation Pharmazeutisher Und Chemisch-Technischer Produkte (Verlag von Julius Springer, Berlin, 1931), p. 364
P.A. Hays, G.S. Remaud, E. Jamin, Y.L. Martin, Geographic origin determination of heroin and cocaine using site-specific isotopic ratio deuterium NMR. J. Forensic Sci. 45, 552–562 (2000)
S. Klemenc, 4-Dimethylaminopyridine as a catalyst in heroin synthesis. Forensic Sci. Int. 129, 194–199 (2002)
T.S. Bailey, P.S. Gee, R. Rezaie, Process for the synthesis of hydromorphone. US WO2006005112A1, PCT/AU2005/001002 (2006)
R. Vardanyan, V. Hruby, Synthesis of Essential Drugs (Elsevier, Amsterdam, 2006), p. 25
M.T. Long, A.M. Hailes, G.W. Kirby, N.C. Bruce, Transformations of morphine alkaloids by Pseudomonas putida M10. Appl. Environ. Microbiol. 61, 3645 (1995)
R. Csuk, G. Vasileva, A. Barthel, Towards an efficient preparation of hydromorphone. Synthesis 44(18), 2840–2842 (2012)
B. Murphy, I. Šnajdr, A. Machara, M.A.A. Endoma-Arias, T.C. Stamatatos, D.P. Cox, T. Hudlický, Conversion of thebaine to oripavine and other useful intermediates for the semisynthesis of opiate-derived agents: synthesis of hydromorphone. Adv. Synth. Catal. 356, 2679–2687 (2014)
L. Rycek, J.J. Hayward, M.A. Latif, J. Tanko, R. Simionescu, T. Hudlicky, Chemoenzymatic total synthesis of hydromorphone by an oxidative dearomatization/intramolecular [4 + 2] cycloaddition sequence: a second-generation approach. J. Org. Chem. 81, 10930–10941 (2016)
V. Varghese, T. Hudlicky, Short chemoenzymatic total synthesis ofent-hydromorphone: an oxidative dearomatization/intramolecular [4+2] cycloaddition/amination sequence. Angew. Chem. 53(17), 4355–4358 (2014)
S.A. Chambers, J.M. DeSousa, E.D. Huseman, S.D. Townsend, The DARK side of total synthesis: strategies and tactics in psychoactive drug production. ACS Chem. Neurosci. 9, 2307–2330 (2018)
T.H. Black, J.C. Forsee, D.A. Probst, A rapid, nearly quantitative conversion of codeine to hydrocodone. Synth. Commun. 30(17), 3195–3201 (2000)
R.J. Carroll, H. Leisch, L. Rochon, T. Hudlicky, D.P. Cox, One-pot conversion of thebaine to hydrocodone and synthesis of neopinone ketal. J. Org. Chem. 74, 747–752 (2009)
T. Hudlicky, V. Varghese, Total synthesis of dihydrocodeine and hydrocodone via a double claisen rearrangement and C-10/C-11 closure strategy. Synlett 24(03), 369–374 (2013)
A.B. Gomez, P. Holmberg, J.E. Backvall, B. Martin-Matute, Transition metal-catalyzed redox isomerization of codeine and morphine in water. RSC Adv. 4, 39519–39522 (2014)
R. Krassnig, C. Hederer, H. Schmidhamme, Optimization of the synthesis of oxycodone and 5-methyloxycodonearch. Pharm. Phan. Med. Chem. 329, 325–326 (1996)
G.B. Kok, P.J. Scammells, Improved synthesis of 14-hydroxy opioid pharmaceuticals and intermediates. RSC Adv. 2, 11318–11325 (2012)
A. Kimishima, H. Umihara, A. Mizoguchi, S. Yokoshima, T. Fukuyama, Synthesis of (−)-oxycodone. Org. Lett. 16(23), 6244–6247 (2014)
T. Hudlicky, M.A. Endoma-Arias, M. Makarova, H. Dela Paz, Chemoenzymatic total synthesis of (+)-oxycodone from phenethyl acetate. Synthesis 51, 225–232 (2018)
A. Lipp, M. Selt, D. Ferenc, D. Schollmeyer, R. Waldvogel, T. Opatz, Synthesis of (–)-oxycodone. Org. Lett. 21, 1828–1831 (2019)
G.I.L. Kang, Synthesis and Applications of Deuterated Methadone and Metabolites to Biotransformation and Disposition Studies. Ph.D. Dissertation, University of British Columbia (1981)
C.J. Barnett, J.C. Smirz, Stereochemistry of bockmuhl’s synthesis of methadone. J. Org. Chem. 41, 710–711 (1976)
A.H. Beckett, N.J. Harper, 162. ConfzgurationaE studies in synthetic analgesics: the synthesis of (-)-methadone from D-( -)-ahnine. J. Chem. Soc. 0(0), 858–861 (1957)
D.L. Hachey, M.J. Kreek, D.H. Mattson, Quantitative analysis of methadone in biological fluids using deuterium-labeled methadone and GLC-chemical-ionization mass spectrometry. J. Pharm. Sci. 66, 1579–1589 (1977)
G.I.L. Kang, F.S. Abbotts, R. Burton, Synthesis and Mass Spectrometry of Deuterated Methadone and Methadone Metabolites. Biomed. Mas. Spectrom. 6, 179–186 (1979)
B.M. Gérardy, J. Poupaert, P. Dumont, An efficient synthesis of the enantiomers of methadone-D10. Bull. Soc. Chim. Belg. 90(9), 977–980 (2010)
C. Alvarado, A. Guzman, E. Diaz, R. Patino, Synthesis of Tramadol and Analogous. J. Mex. Chem. Soc. 49(4), 324–327 (2005)
F. Lecerf-Schmidt, R. Haudecoeur, B. Peres, M.M.F. Queiroz, L. Marcourt, S. Challal, E.F. Queiroz, G.S. Taiwe, T. Lomberget, M.L. Borgne, J.L. Wolfender, M.D. Waard, R.J. Robinsg, A. Boumendjel, Biomimetic synthesis of Tramadol. Chem. Commun. 51(77), 14451–14453 (2015)
S. Sonavane, R. Walavalkar, N. Pradhan, Comparative performance evaluation & systematic screening of 2-Methf as green solvent for cost effective, improved industrial production of tramadol hydrochloride. IJDR 7, 15890–15894 (2017)
G.A.W. Beaudoin, P.J. Facchini, Benzylisoquinoline alkaloid biosynthesis in opium poppy. Planta 240(1), 19–32 (2014)
M. Dastmalchi, M.R. Park, J.S. Morris, P. Facchini, Family portraits: the enzymes behind benzylisoquinoline alkaloid diversity. Phytochem. Rev. 17(2), 249–277 (2017)
J. Ziegler, P.J. Facchini, R. Geißler, J. Schmidt, C. Ammera, R. Kramell, S. Voigtländer, A. Gesell, S. Pienkny, W. Brandt, Evolution of morphine biosynthesis in opium poppy. Phytochemistry 70, 1696–1707 (2009)
Y. Alagoz, T. Gurkok, B. Zhang, T. Unver, Manipulating the biosynthesis of bioactive compound alkaloids for next-generation metabolic engineering in opium poppy using CRISPR-Cas 9 genome editing technology. Sci. Rep. 6(1), 30910 (2016)
A. Onoyovwe, J.M. Hagel, X. Chen, M.F. Khan, D.C. Schriemer, P.J. Facchini, Morphine biosynthesis in opium poppy involves two cell types: sieve elements and laticifers. Plant Cell 25(10), 4110–4122 (2013)
X. Chen, J.M. Hagel, L. Chang, J.E. Tucker, S. Shiigi, Y. Yelpaala, H.Y. Chen, R. Estrada, J. Colbeck, M. Enquist-Newman, A.B. Ibáñez, G. Cottarel, G.M. Vidanes, P.J. Facchini, A pathogenesis-related 10 protein catalyzes the final step in thebaine biosynthesis. Nat. Chem. Biol. 14(7), 738–743 (2018)
F.R. Stermitz, H. Rapoport, The biosynthesis of opium alkaloids, alkaloid interconversions in papaver somniferum and P. orientale. J. Am. Chem. Soc. 83, 4045–4050 (1961)
S. Pathak, D. Lakhwani, P. Gupta, B.K. Mishra, S. Shukla, M.H. Asif, P.K. Trivedi, Comparative transcriptome analysis using high papaverine mutant of papaver somniferum reveals pathway and uncharacterized steps of papaverine biosynthesis. PLoS ONE 8(5), e65622 (2013)
B.H. Novak, T. Hudlicky, J.W. Reed, J. Mulzer, D. Trauner, Morphine synthesis and biosynthesis-an update. Curr. Org. Synth. 4, 343–362 (2000)
S. Pienkny, W. Brandt, J. Schmidt, R. Kramell, J. Ziegler, Functional characterization of a novel benzylisoquinoline O-methyltransferase suggests its involvement in papaverine biosynthesis in opium poppy (Papaversomniferum L). Plant J. 60, 56–67 (2009)
Y. Li, C.D. Smolke, Engineering biosynthesis of the anticancer alkaloid noscapine in yeast. Nat. Commun. 7, 12137–12151 (2016)
Y. Li, S. Li, K. Thodey, I. Trenchard, A. Cravens, C.D. Smolke, Complete biosynthesis of noscapine and halogenated alkaloids in yeast. Proc. Natl. Acad. Sci. 115(17), E3922–E3931 (2018)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kaboudin, B., Sohrabi, M. Chemistry and synthesis of major opium alkaloids: a comprehensive review. J IRAN CHEM SOC 18, 3177–3218 (2021). https://doi.org/10.1007/s13738-021-02268-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-021-02268-y