Thrombophilia.ppt
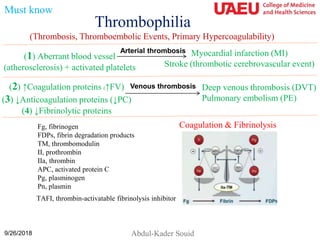
- 1. Thrombophilia 9/26/2018 Abdul-Kader Souid (Thrombosis, Thromboembolic Events, Primary Hypercoagulability) (1) Aberrant blood vessel (atherosclerosis) + activated platelets Myocardial infarction (MI) Stroke (thrombotic cerebrovascular event) Arterial thrombosis (2) ↑Coagulation proteins (↑FV) (3) ↓Anticoagulation proteins (↓PC) (4) ↓Fibrinolytic proteins Venous thrombosis Deep venous thrombosis (DVT) Pulmonary embolism (PE) Must know Fg, fibrinogen FDPs, fibrin degradation products TM, thrombomodulin II, prothrombin IIa, thrombin APC, activated protein C Pg, plasminogen Pn, plasmin TAFI, thrombin-activatable fibrinolysis inhibitor Coagulation & Fibrinolysis
- 2. Arterial Thrombosis Abnormal vessel wall + Activated platelets 2 Arterial thrombosis Myocardial infarction (MI) Thrombotic cerebrovascular event (stroke) Arterial thrombosis = Platelet adhesion to abnormal vessel wall (atherosclerosis) followed by platelet activation, aggregation, and recruitment → MI or stoke 9/26/2018 Must know
- 3. Venous Thrombosis Primary hypercoagulability ↑Coagulation (↑V, ↑ II, ↑ VIII, ↑ IX, ↑ X, ↑ XI) ↓Anticoagulation (↓C, ↓S, ↓Antithrombin) ↓Fibrinolysis Spontaneous clots = Homozygosity or double heterozygosity Precipitated clots (1) ↓Blood flow (pregnancy, surgery, long-haul travel, indwelling catheters, dehydration) (2) ↑Estrogen (oral contraceptives, pregnancy) (3) Obesity, diabetes, aging 3 Venous thrombosis Deep venous thrombosis (DVT) Pulmonary embolism (PE) 9/26/2018 Must know Coagulation & Fibrinolysis
- 4. Fibrinolysis Coagulation and fibrinolysis exist as a fine hemostatic balance. Thrombin (IIa) is the central enzyme in coagulation. It cleaves the soluble fibrinogen (Fg) to insoluble fibrin (FN) clot. Plasmin (PN) is the central enzyme in fibrinolysis. Plasmin is generated from plasminogen (PG) by the activity of tissue plasminogen activator (tPA). Plasmin digests the insoluble fibrin (FN) clot into soluble fibrin degradation products (FDPs). The balance between these two systems is controlled by natural inhibitors of the cascades. Thrombin generation is inhibited by Protein C (PC). Plasmin generation is inhibited by “thrombin-activatable fibrinolysis inhibitor” (TAFI). These two regulatory processes are controlled by the trans-membrane “thrombin- thrombomodulin (II-TM) complex”. Binding of thrombin to thrombomodulin converts its substrate specificity from promoting fibrin formation to inhibiting plasmin generation. Must know Fg, fibrinogen; FDPs, fibrin degradation products; TM, thrombomodulin; II, prothrombin; IIa, thrombin; APC, activated protein C; Pg, plasminogen; Pn, plasmin; TAFI, thrombin-activatable fibrinolysis inhibitor. Coagulation & Fibrinolysis Plasminogen Plasmin Fibrin FDPs tPA (activates) IIa-TM (inhibits)
- 5. Harrison's Principles of Internal Medicine, 18e > Chapter 117. Arterial and Venous Thrombosis Table 117–2 Heritable Causes of Arterial and Venous Thrombosis A. Arterial Thrombosis Platelet Receptors: β3 and α2 integrins PlA2 polymorphism Fc(gamma)RIIA GPIV T13254C polymorphism GPIb Thrombin receptor PAR-1-5061 → D Redox Enzymes: Plasma glutathione peroxidase H2 promoter haplotype Endothelial nitric oxide synthase: −786T/C, −922A/G, −1468T/A Paraoxonase: −107T allele, 192R allele Homocysteine: Cystathionine β-synthase 833T → C 5,10-methylene tetrahydrofolate reductase (MTHFR) 677C → T B. Venous Thrombosis Coagulant Proteins: Fibrinogen: −455G/A, −854G/A Prothrombin (20210G → A) Protein C Anticoagulant Pathway Factor V Leiden: 1691G → A (Arg506Gln) Thrombomodulin: 1481C → T (Ala455Val) Fibrinolytic Proteins with Known Polymorphisms: Tissue plasminogen activator (tPA): 7351C/T, 20 099T/C in exon 6, 27 445T/A in intron 10 Plasminogen activator inhibitor (PAI-1): 4G/5G insertion/deletion polymorphism at position −675 9/26/2018 Optional No need to routinely test for: Homocysteine, factor VIII, MHTFR / XIII polymorphism, & PAI-I
- 6. ↑Coagulation Proteins ↑Factor V half-life (mutations in factor V causing resistance to proteolysis by protein C (protein C resistance) ↑Factor II (Prothrombin mutation G20210A) ↑Factor VIII, IX, X, XI 6 ↓Anticoagulation Proteins ↓Protein C ↓Protein S ↓Antithrombin (strongest risk) Heritable Thrombosis (short list) 9/26/2018 ↓Plasminogen ↓Fibrinolytic Proteins Must know ↑Factor V half-life (important) The screening test is “activated protein C resistance (APC resistance)” Factor V mutations; eg, Factor V Leiden (= resistant factor V) = G1691A (Arg506Gln) ↑Homocysteine - MTHFR polymorphism → arterial thrombosis (weak effect; no need for testing) No need to routinely test for: Homocysteine, factor VIII, MHTFR / XIII polymorphism, & PAI-I
- 7. Activated Protein C (APC) Resistance Assay (An aPTT-based Assay) Activated partial thromboplastin time (aPTT) Citrated plasma + phospholipid + Ca2+ + silica → Measure time to fibrin clot Citrated plasma + phospholipid + Ca2+ + silica + Activated protein C [APC] → ↑Time to fibrin clot (a normal response to APC) Citrated plasma + phospholipid + Ca2+ + silica + Activated protein C [APC} → No change in the time to form fibrin clot (an abnormal response to APC = APC Resistance) Protein C inactivates factor V. Factor V Mutations (e.g., Factor V Leiden) • They are the most common inherited risk factor for thrombosis. • Mutation causes factor V to become resistant to proteolysis by protein C. • It affects 5% of the U.S. white population (heterozygous state). • Individuals who are heterozygous have a 5-fold ↑risk of venous thrombosis (especially women who are taking oral contraceptives). • Individuals who are homozygotes have a 100-fold ↑risk of venous thrombosis. Must know
- 8. Protein C Deficiency The anticoagulant Protein C is activated by a thrombin- thrombomodulin (IIa-TM) complex on the surface of endothelial cells. Activated protein C (APC) then binds to protein S and the resulting protease inactivates factor V, halting thrombin generation. Homozygous Protein C deficiency presents early in infancy with purpura fulminans (shown), which is treated with daily protein C (plasma for protein S deficiency). 9/26/2018 8 Must know
- 9. A More Complete Work-up of Thrombosis 9 1. Activated protein C (APC) resistance assay (aPTT-based assay) 2. Factor V Leiden (DNA-based assay, G1691A) 3. Factor II 20210G → A (prothrombin mutation) 4. Protein C & S activity and antigen 5. Antithrombin activity & antigen 6. Homocysteine (blood level) 7. Methyltetrahydrofolate reductase (MTHFR) genetic testing (if ↑homocysteine) 8. Plasminogen activity 9. Tissue plasminogen activator (TPA) antigen 10. Plasminogen activator inhibitor activity (PAI-1) 11. α2–Antiplasmin activity 12. Lipoprotein A (decreases fibrinolysis) 9/26/2018 Optional
- 10. Vitamin K & Warfarin • Vitamin K is a fat-soluble vitamin, required for complete synthesis of factors II (prothrombin), VII, IX, X, Protein C, and Protein S. These six proteins require vitamin K for the addition of 9-12 γ-carboxyl groups (COO-) at specific glutamate residues. The resulting carboxylated glutamates mediate Ca2+-dependent binding of these factors to platelet surface (phospholipids). • Vitamin K deficiency is caused by (1) Diet short of animal fats (egg yolks, milk, red meat) and dark green vegetable, (2) Fat malabsorption (biliary obstruction [cholestasis, direct hyperbilirubinemia], cystic fibrosis), (3) Use of broad-spectrum antibiotics. Vitamin K deficiency is associated with ↑PT/INR, ↑aPTT, and normal TT. • Warfarin (coumarin or Coumadin; an anticoagulant) is vitamin K antagonist, which inhibits the six vitamin K-dependent factors. • The use of warfarin is challenging because the therapeutic range is narrow (INR = 2.0 to 3.0) and dosing is affected by genetic variation, drug interactions, and diet. Time spent with a PT/INR above the therapeutic range increases the risk of bleeding, and time spent below the therapeutic range increases the risk of thromboembolic complications. Warfarin has a large clinical experience and is highly effective in reducing the risk of venous and arterial thromboemboli. Must know
- 11. Warfarin • Factor X is the most potent activator of prothrombin and it is the primary target of anticoagulation by warfarin. Depletion of factor X prevents prothrombin activation. • The half-life of factor X is 20-40 h, shorter than that of prothrombin (60 h). As a result, it takes a few days before the anticoagulant effects (prothrombin depletion) of warfarin are fully achieved. • Thus, for treatment of deep vein thrombosis (DVT), heparin must be overlapped with warfarin (time to effect = 2-5 days; half-live about 40 h) for a few days to maintain anticoagulation. • Reversal of warfarin effect in symptomatic patients include vitamin K (non- urgent reversal; IV, SC, or oral) and Prothrombin Complex Concentrate (II, VII, IX, X; for urgent reversal) + vitamain K. Must know
- 12. Antithrombotic Agents Thrombolysis (recombinant tissue plasminogen activator, rTPA) Unfractionated Heparin Low Molecular Weight Heparin (enoxaparin) Mechanism rTPA + Plasminogen → Plasmin → ↑fibrinolysis Antithrombin– dependent Inactivate Factor X Indication Threatening thrombi Other thrombi Other thrombi Dose IV infusion or injection directly into the thrombus IV bolus followed by infusion Subcutaneous every 12 to 24 h Monitoring ↑D-dimer ↑aPTT Anti-factor X activity Risk of bleeding Medium-to-High Low-to-Medium Low Must know Warfarin has a large clinical experience and is highly effective in treating thrombotic events and in reducing the risk of venous and arterial thromboemboli. The treatment is followed by INR. Contraindications to thrombolysis include (1) Active bleeding; (2) Intracranial neoplasm; (3) Major trauma or surgery within 10 days; (4) Severe hypertension; (5) Active seizure; (6) Prematurity (<32 weeks’ gestation); and (7) Platelet count <50 x109/L or fibrinogen <100 mg/dL (2.94 µmol/L).
- 13. Must Know Pearls • Activated platelets cause arterial thrombosis. • ↑Coagulation factor(s) cause venous thrombosis (e.g., ↑factor V, such as factor V Leiden). – Factor V Leiden = factor V mutation causing resistance to proteolysis by protein C (Protein C resistance). • ↓Anticoagulation factor causes venous thrombosis (e.g., ↓Protein C or S). • ↓Fibrinolytic factor cause venous thrombosis (e.g., ↓plasminogen). • Antithrombotic therapies include ‘recombinant tissue plasminogen activator’ (rTPA), warfarin, unfractionated heparin, and low-molecular weight heparin (enoxaparin, inhibits factor X). • Avoid oral contraceptives in high-risk thrombotic conditions (positive history of blood clots). • Patients with a known risk of thrombosis who are going to be immobilized should receive prophylactic enoxaparin.
- 14. Q. A 17-year-old girl presents with left popliteal deep vein thrombosis (DVT) while taking an estrogen-containing oral contraceptive. Her maternal family history is positive for DVT. Which one of the following conditions is the most likely cause of her precipitated DVT? A. Protein C deficiency B. Prothrombin mutation G20210A C. Antithrombin deficiency D. Plasminogen deficiency E. Factor V Leiden (G1691A; Arg506Gln) 9/26/2018 14
- 15. Required Reading 1. Monagle P, Chan AKC, Goldenberg NA, Ichord RN, Journeycake JM, Nowak-Göttl U, Vesely SK. Antithrombotic therapy in neonates and children: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence- Based Clinical Practice Guidelines. Chest. 2012;141:e737S-e801S. doi: 10.1378/chest.11-2308. 2. Harrison's Principles of Internal Medicine, 18e > Chapter 117. Arterial and Venous Thrombosis 3. Young G, Albisetti M, Bonduel M, et al: Impact of inherited thrombophilia on venous thromboembolism in children: a systematic review and meta-analysis of observational studies. Circulation 118:1373–1382, 2008. 4. Daví G, Patrono C: Platelet activation and atherothrombosis. N Engl J Med 13;357:2482, 2007. 5. Mechanisms of thrombus formation. N Engl J Med 28;359:938, 2008. 6. Tapson VF: Acute pulmonary embolism. N Engl J Med 6;358:1037, 2008. 9/26/2018 15
- 16. Connors JM. Thrombophilia Testing and Venous Thrombosis. N Engl J Med. 2017;377:2298. doi: 10.1056/NEJMc1713797. 9/26/2018 16
- 17. 9/26/2018 17 Connors JM. Thrombophilia Testing and Venous Thrombosis. N Engl J Med. 2017;377:2298. doi: 10.1056/NEJMc1713797.