Answer
383.1k+ views
Hint: Refer to the Goldstein experiment of canal rays. Goldstein was the early investigator of discharge tubes and also the discoverer of the canal rays, also called anode rays. Recall the results of his experiment.
Complete answer:
Canal ray experiment performed by the German scientist Eugen Goldstein in 1886. This experiment led to the discovery of protons. This discovery happened just after the discovery of electrons. In the experiment, Goldstein applied high voltage across a discharge tube which had a perforated cathode. When the voltage was increased to several thousand volts, a faint luminous ray or a faint red glow was observed from the holes behind the cathode on a zinc sulphide screen. These rays were named canal rays, also called anode rays (carrying positive charge particles). The experiment is depicted below:
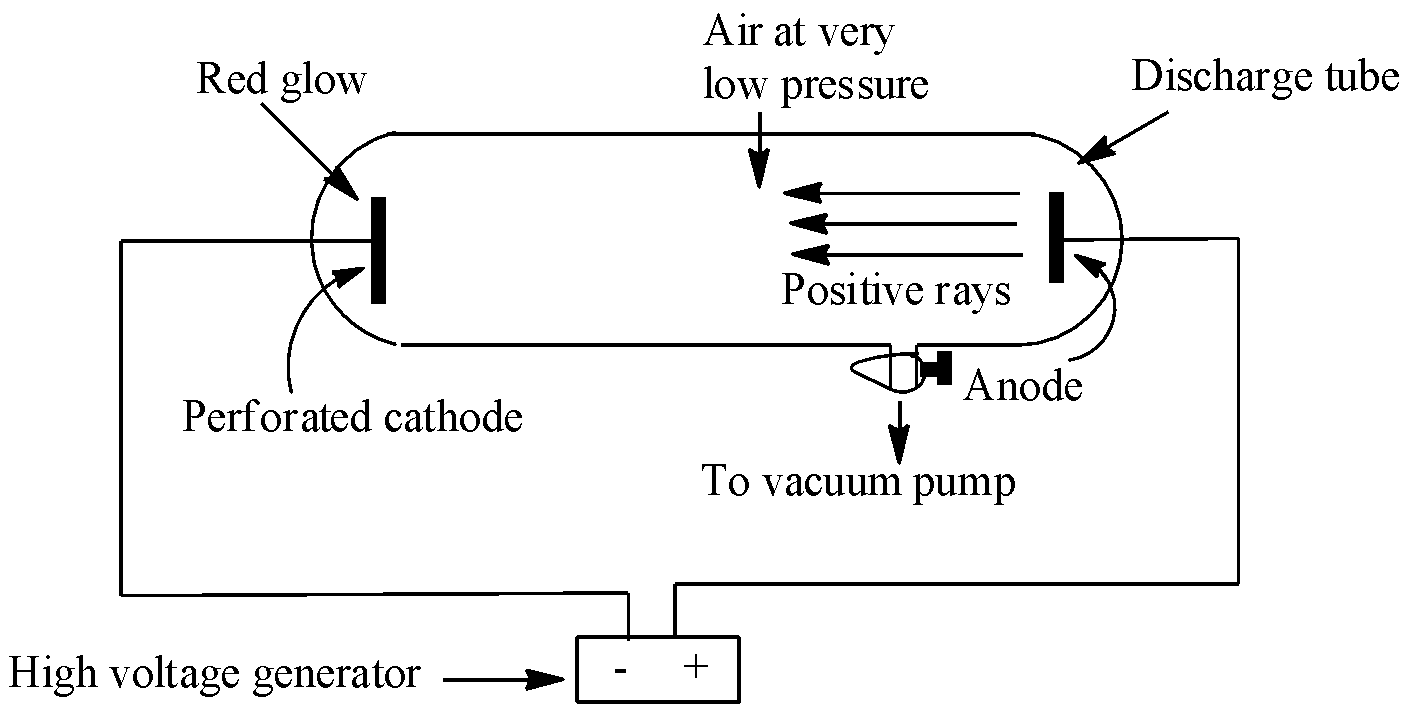
The characteristic of these canal rays are listed below:
- Canal rays are made up of positively charged particles
- In the absence of electrical and magnetic fields, these rays travel in a straight line from anode to cathode.
- The charge to mass ratio or e/m ratio of the canal rays is found to depend on the nature of gas i.e, not same for all gases.
- They produce fluorescence on the ZnS screen.
Hence, among all the given options, the statement given in option C is not the characteristic of canal rays.
Thus, option C is the answer.
Note:
Goldstein also took his own investigations of discharge tubes and cathode rays. He discovered several important properties of cathode rays, which later contributed to the discovery of electrons. Cathode rays travel from negatively charged cathode towards the positively charged anode while anode rays travel in the opposite direction.
Complete answer:
Canal ray experiment performed by the German scientist Eugen Goldstein in 1886. This experiment led to the discovery of protons. This discovery happened just after the discovery of electrons. In the experiment, Goldstein applied high voltage across a discharge tube which had a perforated cathode. When the voltage was increased to several thousand volts, a faint luminous ray or a faint red glow was observed from the holes behind the cathode on a zinc sulphide screen. These rays were named canal rays, also called anode rays (carrying positive charge particles). The experiment is depicted below:

The characteristic of these canal rays are listed below:
- Canal rays are made up of positively charged particles
- In the absence of electrical and magnetic fields, these rays travel in a straight line from anode to cathode.
- The charge to mass ratio or e/m ratio of the canal rays is found to depend on the nature of gas i.e, not same for all gases.
- They produce fluorescence on the ZnS screen.
Hence, among all the given options, the statement given in option C is not the characteristic of canal rays.
Thus, option C is the answer.
Note:
Goldstein also took his own investigations of discharge tubes and cathode rays. He discovered several important properties of cathode rays, which later contributed to the discovery of electrons. Cathode rays travel from negatively charged cathode towards the positively charged anode while anode rays travel in the opposite direction.
Recently Updated Pages
Basicity of sulphurous acid and sulphuric acid are

Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

What is the stopping potential when the metal with class 12 physics JEE_Main

The momentum of a photon is 2 times 10 16gm cmsec Its class 12 physics JEE_Main

How do you arrange NH4 + BF3 H2O C2H2 in increasing class 11 chemistry CBSE

Is H mCT and q mCT the same thing If so which is more class 11 chemistry CBSE

Trending doubts
Difference Between Plant Cell and Animal Cell

Difference between Prokaryotic cell and Eukaryotic class 11 biology CBSE

Fill the blanks with the suitable prepositions 1 The class 9 english CBSE

Change the following sentences into negative and interrogative class 10 english CBSE

How many millions make a billion class 6 maths CBSE

Fill the blanks with proper collective nouns 1 A of class 10 english CBSE

Give 10 examples for herbs , shrubs , climbers , creepers

What organs are located on the left side of your body class 11 biology CBSE

What is BLO What is the full form of BLO class 8 social science CBSE



