Abstract
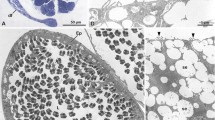
The clear phylogenetic status of the enigmatic Phylum Dicyemida is still uncertain. Their primitive body plan lacks essential metazoan synapomorphies, while genetic data favor a kinship with higher lophotrochozoans. This ultrastructural study increases the confusion about this phylum by presenting an unusual gonad and sperm structure lacking all synapomorphies essential for the various phyla of the lophotrochozoans, either free-living ones or parasites. In Dicyema typus, gonadogenesis is reduced to a single somatic cell, i.e., the infusorigen’s axial cell that functions as a somatic gonadal founder cell as soon as a spermatogonium takes residence in its cytoplasm. The spermiogenic cells resulting therefrom are not connected by intercellular bridges and permanently contain a bundle of microtubules in their cytoplasm, obviously a kind of “dormant” spindle having assembled without centrosomes. Primary spermatocytes develop so-called polycomplexes, multiple synaptinemal complexes. The structure of the sperm is based on a certain kind of somatic cell that has been minimally adapted to function as a sperm. The mature sperm consists in only three organelles: an ovoid nucleus with a somatic chromatin structure, numerous pore complexes and a centrally arranged cluster of coiled tubular structures; a bundle of microtubules embedded into a rim running along the nucleus longitudinal surface, projecting out of the cell like a spear; and a lipid vesicle tightly attached to one pole of the nucleus, touching there the adjacent bundle of microtubules. Immunelectron microscopy confirms the somatic condition of mature sperm, revealing somatic histone H1 immunoreactivity over the nucleus, which can be interpreted as synapomorphy shared with Protozoa, Porifera and Cnidaria. Fertilization occurs as selfing, where the sperm penetrates, “bundle of microtubules first”, a primary oocyte attached vis-à-vis on the other side of the plasma membrane of the infusorigen’s axial cell. This somatic situation points to an ancient evolutionary model rather than to a condition caused by retrogression due to their life as commensals.




Similar content being viewed by others
References
Ausio J (1995) Histone H1 and the evolution of the nuclear sperm-specific proteins. In: Jamieson BGM, Ausio J, Justine JL (eds) Advances in Spermatozoal Phylogeny and Taxonomy. Mem Mus Natl Hist Nat, Paris, pp 447–462
Ausio J, vanVeghel MLJ, Gomez R, Barreda D (1997) The sperm nuclear basic proteins (SNBPs) of the sponge Neofibularia nolitangere. Implications for the molecular evolution of SNBPs. J Mol Evol 45:91–96
Baccetti B, Afzelius B (1976) The biology of the sperm cell. Karger, Basel
Beneden van E (1882) Contribution a l’histoire des Dicyémides. Arch Biol 3:195–228
Bresciani J, Fenchel T (1965) Studies on dicyemid Mesozoa. I the fine structure of the adult (the nematogen and rhombogen stage). Vidensk Medd Dan Naturhist Foren 128:85–92
Cavalier-Smith T (1993) Kingdom protozoa and its 18 phyla. Microbiol Rev 58:953–994
Czaker R (2000) Extracellular matrix (ECM) components in a very primitive multicellular animal, the dicyemid mesozoan Kantharella antarctica. Anat Rec 259:52–59
Dallai R, Mazzini M (1983) Spermatozoa and diptera phylogeny. In: Andrè J (ed) The sperm cell. Martinus Nijhoff, The Hague, pp 446–450
Dodson EO (1956) A note on the systematic position of the mesozoa. Syst Zool 5:37–40
Franzén A (1956) On spermiogenesis, morphology of the spermatozoon and biology of fertilization among invertebrates. Zool Bidr Upps 31:335–482
Franzén A (1970) Phylogenetic aspects of the morphology of the spermatozoan and spermio-genesis. In: Baccetti B (ed) Comparative spermatology. Academic, New York, pp 29–46
Furuya H, Tsuneki G, Koshida Y (1993) The development of the hermaphroditic gonad in four species of dicyemid mesozoans. Zool Sci 10:455–466
Furuya H, Tsuneki G, Koshida Y (1996) The cell lineages of two types of embryos and hermaphroditic gonad in dicyemid mesozoans. Dev Growth Differ 38:453–463
Furuya H, Tsuneki K, Koshida Y (1997) Fine structure of dicyemid mesozoans with special reference to cell junctions. J Morphol 231:297–305
Goffredo S, Teló T, Scanabissi F (2000) Ultrastructural observations of the spermatogenesis of the hermaphroditic, solitary coral balanophyllia europaea (Anthozoa, Scleractinia). Zoomorphology 119:231–240
Goldstein P (1987) Multiple synaptinemal complexes (polycomplexes): origin, structure and function. Cell Biol Int Rep 11:759–796
Guo GQ, Zheng GC (2004) Hypotheses for functions of intercellular bridges in male germ cell development and its cellular mechanisms. J Theor Biol 229:139–146
Heald R, Tournebize R, Blank T, Sandaltzopoulos R, Becker R, Hyman A, Karsenti E (1996) Self-organization of microtubules into bipolar spindles around artificial chromosomes in Xenopus egg extracts. Nature 382:420–425
Hinsch GW (1974) Comparative ultrastructure of cnidarian sperm. Am Zool 14:457–465
Hori H, Osawa S (1987) Origin and evolution of organisms as deduced from 5 S ribosomal RNA sequences. Mol Biol Evol 4:445–472
Hyman LH (1940) The invertebrates, vol 1. McGraw-Hill, New York
Jamieson BGM (1987) The ultrastructure and phylogeny of insect spermatozoa. Cambridge University Press, Cambridge
Jamieson BGM, Ausio J, Justine JL (1995) Advances in spermatozoal phylogeny and taxonomy. Mem Mus Natl Hist Nat, Paris
Justine JL (1995) Spermatozoal ultrastructure and phylogeny in the parasitic Platyhelminthes. In: Jamieson BGM, Ausio J, Justine JL (eds) Advances in spermatozoal phylogeny and taxonomy. Mem Mus Natl Hist Nat, Paris, pp 55–86
Kasinski HE (1995) Evolution and origin of sperm nuclear basic proteins. In: Jamieson BGM, Ausio J, Justine JL (eds) Advances in spermatozoal phylogeny and taxonomy. Mem Mus Natl Hist Nat, Paris, pp 463–473
Katayama T, Wada H, Furuya H, Satoh N, Yamamoto M (1995) Phylogenetic position of the dicyemid mesozoa inferred from 18S rDNA sequences. Biol Bull 189:81–90
Kessel RG (1992) A last frontier in cellular organelles. Int Rev Cytol 133:43–120
Kobayashi M, Furuya H, Holland PWH (1999) Dicyemids are higher animals. Nature 401:762
Kobayashi M, Furuya H, Wada H (2009) Molecular marker compairing the extremely simple body plan of dicyemids to that of lophotrochozoans: insight from the expression patterns of Hox, Otx and brachyury. Evol Dev 11:582–589
Lapan EA, Morowitz HJ (1972) The Mesozoa. Sci Am 222:94–101
Matsubara JA, Dudley PL (1976) Fine structural studies of the dicyemid mesozoan Dicyemmenea californica McConnaughey. J Parasitol 62:377–389
McConnaughey BH (1951) The life cyle of the dicyemid mesozoa. Univ Calif Publ Zool 55:295–336
Morales CR, Wu XQ, Hecht NB (1998) The DNA/RNA binding protein TB-RBP moves from the nucleus to the cytoplasm and through intercellular bridges in male germ cells. Dev Biol 201:113–123
Noto T, Endoh H (2004) A ‘chimera’ theory on the origin of dicyemid mesozoans: evolution driven by frequent lateral gene transfer from host to parasite. Biosystems 73:73–83
Noto T, Yazaki K, Endoh H (2003) Developmentally regulated extrachromosomal circular DNA formation in the mesozoan Dicyema japonicum. Chromosoma 111:359–368
Nouvel H (1947) Les Dicyémids. Arch Biol 58:54–220
Ogino K, Tsuneki K, Furuya H (2010) Unique genome of dicyemid mesozoan: Highly shortened spliceosomal introns in conservative exon/intron structure. Gene 449:70–76
Ohama T, Kumazaki K, Hori H, Osawa S (1984) Evolution of multicellular animals as deduced from 5S ribosomal RNA-squences: a possible early emergence of the Mesozoa. Nucleic Acids Res 12:5101–5108
Paulus W (1989) Ultrastructural investigation of spermatogenesis in Spongilla lacustris and Ephydatia fluviatilis (Porifera, Spongillidae). Zoomorphology 109:123–130
Pawlowski J, Montoya-Burgos JI, Fahrni FJ, Wüest J, Zaninetti L (1996) Origin of the mesozoa inferred from 18S rRNA gene sequences. Mol Biol Evol 13:1128–1132
Ridley RK (1969) Electron microscopic studies on dicyemid mesozoa. II Infusorigen and infusoriform stages. J Parasitol 55:779–793
Rizzo PJ, Bradley W, Morris RL (1985) Histones of the unicellular algae Olisthodiscus luteus. Biochemistry 24:1727–1732
Rocchini C, Marx MR, von Carosfeld JS, Kasinsky HE, Rosenberg E, Sommer F, Ausio J (1996) Replacement of nucleosomal histones by histone H1-like proteins during spermiogenesis in Cnidaria: evolutionary implications. J Mol Evol 42:240–246
Short RB, Damian RT (1967) Oogenesis, fertilization, and first cleavage of Dicyema aegira. J Parasitol 53:186–195
Steffen W, Fuge H, Dietz R, Bastmeyer M, Muller G (1986) Aster-free spindle poles in insect spermatocytes, evidence for chromosome induced spindle formation? J Cell Biol 102:1679–1867
Steiner SSC (1991) Sperm morphology of scleractinians from the Caribbean. Hydrobiologia 216(217):131–135
Stunkard HW (1954) The life history and systematic relations of the mesozoa. Q Rev Biol 29:230–244
Subirana JA, Cozcolluela C, Palau J, Unzeta M (1973) Protamines and other basic proteins from spermatozoan of molluscs. Biochim Biophys Acta 317:364–379
Sym M, Roeder GS (1995) Zip 1-induced changes in synaptonemal complex structure and polycomplex assembly. J Cell Biol 128:455–466
Vasquez GH, Chavez B, Martin CT (1973) A preferential staining method for chromatin in electron microscopy. J Microsc 16:243–246
Acknowledgements
The author thanks Ms. Marianne Steiner and Ms. Erika Vanyek for their skilled technical assistance and Ms. Christa Farrenkopf for her competent editorial cooperation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Czaker, R. Dicyemid’s dilemma: structure versus genes. The unorthodox structure of dicyemid reproduction. Cell Tissue Res 343, 649–658 (2011). https://doi.org/10.1007/s00441-010-1124-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-010-1124-z