Abstract
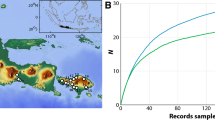
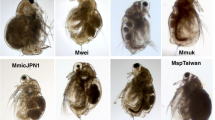
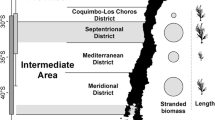
The underexplored intertidal ecosystems of Antarctica are facing rapid changes in important environmental factors. Associated with temperature increase, reduction in coastal ice will soon expose new ice-free areas that will be colonized by local or distant biota. To enable detection of future changes in faunal composition, a biodiversity baseline is urgently required. Here, we evaluated intertidal faunal diversity at 13 locations around the Gerlache Strait (western Antarctic Peninsula), using a combination of a quadrat approach, morphological identification and genetic characterization. Our data highlight a community structure comprising four generally distributed and highly abundant species (the flatworm Obrimoposthia wandeli, the bivalve Kidderia subquadrata, and the gastropods Laevilitorina umbilicata and Laevilitorina caliginosa) as well as 79 rarer and less widely encountered species. The most abundant species thrive in the intertidal zone due to their ability to either survive overwinter in situ or to rapidly colonize this zone when conditions allow. In addition, we confirmed the presence of multiple trophic levels at nearly all locations, suggesting that complex inter-specific interactions occur within these communities. Diversity indices contrasted between sampling locations (from 3 to 32 species) and multivariate approaches identified three main groups. This confirms the importance of environmental heterogeneity in shaping diversity patterns within the investigated area. Finally, we provide the first genetic and photographic baseline of the Antarctic intertidal fauna (106 sequences, 137 macrophotographs), as well as preliminary insights on the biogeography of several species. Taken together, these results provide a timely catalyst to assess the diversity and to inform studies of the potential resilience of these intertidal communities.



Similar content being viewed by others
Data availability
All genetic data, as well as photographs of voucher specimens, can be publicly accessed and downloaded on boldsystems.org, under the “INTGS” project (Intertidal fauna from the Gerlache Strait). Occurrence data can be found on the Global Biodiversity Information Facility (GBIF, B121 dataset) using the following link: https://doi.org/10.15468/56bv6z. Additional data, tables and figures are also provided as Supplementary materials. The specimens used in this study are hosted at the Université Libre de Bruxelles (ULB, Brussels, Belgium) – Laboratoire de Biologie Marine.
References
Adami, M. L., & Gordillo, S. (1999). Structure and dynamics of the biota associated with Macrocystis pyrifera (Phaeophyta) from the Beagle Channel. Tierra del Fuego. Scientia Marina, 63(S1), 183–191. https://doi.org/10.3989/scimar.1999.63s1183
Aghmich, A., Taboada, S., Toll, L., & Ballesteros, M. (2016). First assessment of the rocky intertidal communities of Fildes Bay, King George Island (South Shetland Islands, Antarctica). Polar Biology, 39(1), 189–198. https://doi.org/10.1007/s00300-015-1814-9
Arnaud, P. M. (1974). Contribution à la bionomie marine benthique des régions antarctiques et subantarctiques. Station Marine d'Endoume, 32 3pp.
Barnes, D. K., & Arnold, R. J. (2003). Possible latitudinal clines in Antarctic intertidal and subtidal zone communities encrusting ephemeral hard substrata. Journal of Biogeography, 26(2), 207–213. https://doi.org/10.1046/j.1365-2699.1999.00264.x
Barnes, D. K., Kaiser, S., Griffiths, H. J., & Linse, K. (2009). Marine, intertidal, freshwater and terrestrial biodiversity of an isolated polar archipelago. Journal of Biogeography, 36(4), 756–769. https://doi.org/10.1111/j.1365-2699.2008.02030.x
Bick, A., & Arlt, G. (2013). Description of intertidal macro-and meiobenthic assemblages in Maxwell Bay, King George Island, South Shetland Islands. Southern Ocean. Polar Biology, 36(5), 673–689. https://doi.org/10.1007/s00300-013-1293-9
Borcard, D., Gillet, F., & Legendre, P. (2018). Numerical Ecology with R. 452p. Springer.
Brasier, M. J., Barnes, D. K., Bax, N., Brandt, A., Christianson, A. B., Constable, A. J., & Waller, C. L. (2021). Responses of Southern Ocean seafloor habitats and communities to global and local drivers of change. Frontiers in Marine Science, 8, 109. https://doi.org/10.3389/fmars.2021.622721
Brasier, M. J., Wiklund, H., Neal, L., Jeffreys, R., Linse, K., Ruhl, H., & Glover, A. G. (2016). DNA barcoding uncovers cryptic diversity in 50% of deep-sea Antarctic polychaetes. Royal Society Open Science, 3(11), 160432. https://doi.org/10.1098/rsos.160432
Chao, A., Ma, K. H., Hsieh, T. C., & Chiu, C. H. (2015). Online program SpadeR (Species-richness prediction and diversity estimation in R). Program and User’s Guide published at http://chao.stat.nthu.edu.tw/wordpress/software_download
Chelchowski, M., Balazy, P., & Kuklinski, P. (2022). Seasonal variability in macrobenthos assemblage parameters in the highly disturbed Antarctic intertidal zone–Relatively rich biodiversity year around. Estuarine, Coastal and Shelf Science, 278, 108114. https://doi.org/10.1016/j.ecss.2022.108114
Christiansen, H., Dettai, A., Heindler, F. M., Collins, M. A., Duhamel, G., Hautecoeur, M., & Van de Putte, A. P. (2018). Diversity of mesopelagic fishes in the Southern Ocean-a phylogeographic perspective using DNA barcoding. Frontiers in Ecology and Evolution, 120. https://doi.org/10.3389/fevo.2018.00120
Clark, G. F., Stark, J. S., & Johnston, E. L. (2017). Tolerance rather than competition leads to spatial dominance of an Antarctic bryozoan. Journal of Experimental Marine Biology and Ecology, 486, 222–229. https://doi.org/10.1016/j.jembe.2016.10.008
Clarke, A., & Beaumont, J. C. (2020). An extreme marine environment: a 14-month record of temperature in a polar tidepool. Polar Biology, 43(12), 2021–2030. https://doi.org/10.1007/s00300-020-02762-8
Convey, P. (2010). Terrestrial biodiversity in Antarctica-Recent advances and future challenges. Polar Science, 4(2), 135–147. https://doi.org/10.1016/j.polar.2010.03.003
Convey, P., & Peck, L.S. (2019). Antarctic environmental change and biological responses. Science Advances, 5(11), eaaz0888. https://doi.org/10.1126/sciadv.aaz0888
Costello, M. J., Wilson, S., & Houlding, B. (2013). More taxonomists describing significantly fewer species per unit effort may indicate that most species have been discovered. Systematic Biology, 62(4), 616–624. https://doi.org/10.1093/sysbio/syt024
Danis, B., Christiansen, H., Guillaumot, C., Heindler, F., Houston, R., Jossart, Q., & Saucède, T. (2019). Report of the Belgica 121 expedition to the West Antarctic Peninsula. 96 pp.
Danis, B., Christiansen, H., Guillaumot, C., Heindler, F., Jossart, Q., Moreau, C., & Saucède, T. (2021). The Belgica 121 expedition to the Western Antarctic Peninsula: a high resolution biodiversity census. Biodiversity Data Journal, 9, e70590. https://doi.org/10.3897/BDJ.9.e70590
Danis, B., Wallis, B., Guillaumot, C., Moreau, C., Pasotti, F., & Saucède, T. (2022). Nimble vessel cruises as an alternative for Southern Ocean biodiversity research: concept and preliminary results from the Belgica121 expedition. Antarctic Science, 34(4): 336-342. https://doi.org/10.1017/S0954102022000165
Dayrat, B. (2005). Towards integrative taxonomy. Biological journal of the Linnean Society, 85(3), 407–417. https://doi.org/10.1111/j.1095-8312.2005.00503.x
De Broyer, C., Koubbi, P., Griffiths, H. J., Raymond, B., d’Udekem d’Acoz, C., & Ropert-Coudert, Y. (2014). Biogeographic Atlas of the Southern Ocean.. SCAR, Cambridge. pp 510.
De Deckker, P. (2018). On the long-ignored scientific achievements of the Belgica expedition 1897–1899. Polar Research, 37(1), 1474695.
Dewart, G. (1972). A Magnetic Profile in the Gerlache Strait, Antarctica. Institute of Polar Studies Miscellaneous Series M-83, Research Foundation, The Ohio State University, pp 8.
Dragani, W. C., Drabble, M. R., D’Onofrio, E. E., & Mazio, C. A. (2004). Propagation and amplification of tide at the Bransfield and Gerlache Straits, northwestern Antarctic Peninsula. Polar Geosciences, 17, 156-170.
Dray, S., Bauman, B., Blanchet, G., Borcard, D., Clappe, S., & Wagner, H. (2021). R package ‘adespatial’. https://github.com/sdray/adespatial
Eastman, J. T. (1993). Antarctic fish biology: evolution in a unique environment. Academic Press. pp 322.
Erpenbeck, D., Knowlton, A. L., Talbot, S. L., Highsmith, R. C., & Van Soest, R. W. M. (2004). A molecular comparison of Alaskan and North East Atlantic Halichondria panicea (Pallas 1766) (Porifera: Demospongiae) populations. BMIB-Bollettino dei Musei e degli Istituti Biologici, 68, 319–325.
Figuerola, B., Gore, D. B., Johnston, G., & Stark, J. S. (2019). Spatio-temporal variation of skeletal Mg-calcite in Antarctic marine calcifiers. Plos One, 14(5), e0210231. https://doi.org/10.1371/journal.pone.0210231
Figuerola, B., Griffiths, H. J., Krzeminska, M., Piwoni‐Piorewicz, A., Iglikowska, A., & Kuklinski, P. (2023). Temperature as a likely driver shaping global patterns in mineralogical composition in bryozoans: implications for marine calcifiers under global change. Ecography, e06381. https://doi.org/10.1111/ecog.06381
Figuerola, B., Hancock, A. M., Bax, N., Cummings, V. J., Downey, R., & Stark, J. S. (2021). A review and meta-analysis of potential impacts of ocean acidification on marine calcifiers from the Southern Ocean. Frontiers in Marine Science, 29, 584445. https://doi.org/10.3389/fmars.2021.584445
Fraser, C. I., Morrison, A. K., Hogg, A. M., Macaya, E. C., & Waters, J. M. (2018). Antarctica’s ecological isolation will be broken by storm-driven dispersal and warming. Nature Climate Change, 8(8), 704-708. https://doi.org/10.1038/s41558-018-0209-7
Folmer, O., Black, M., Hoeh, W., Lutz, R., & Vrijenhoek, R. (1994). DNA primers for amplification of mitochondrial cytochrome c oxidase subunit I from diverse metazoan invertebrates. Molecular Marine Biology and Biotechnology, 3, 294–299.
Galera, H., Chwedorzewska, K. J., Korczak-Abshire, M., & Wódkiewicz, M. (2018). What affects the probability of biological invasions in Antarctica? Using an expanded conceptual framework to anticipate the risk of alien species expansion. Biodiversity and Conservation, 27(8), 1789–1809. https://doi.org/10.1007/s10531-018-1547-5
Garcıa, M. A., Castro, C. G., Rios, A. F., Doval, M. D., Rosón, G., Gomis, D., & López, O. (2002). Water masses and distribution of physico-chemical properties in the Western Bransfield Strait and Gerlache Strait during Austral summer 1995/96. Deep Sea Research Part II: Topical Studies in Oceanography, 49(4–5), 585–602. https://doi.org/10.1016/S0967-0645(01)00113-8
González-Wevar, C. A., Gérard, K., Rosenfeld, S., Saucède, T., Naretto, J., Díaz, A., & Poulin, E. (2019). Cryptic speciation in Southern Ocean Aequiyoldia eightsii (Jay, 1839): mio-pliocene trans-drake passage separation and diversification. Progress in Oceanography, 174, 44-54. https://doi.org/10.1016/j.pocean.2018.09.004
Griffiths, H. J., & Waller, C. L. (2016). The first comprehensive description of the biodiversity and biogeography of Antarctic and Sub-Antarctic intertidal communities. Journal of Biogeography, 43(6), 1143–1155. https://doi.org/10.1111/jbi.12708
Griffiths, H. J., Meijers, A. J., & Bracegirdle, T. J. (2017). More losers than winners in a century of future Southern Ocean seafloor warming. Nature Climate Change, 7(10), 749–754. https://doi.org/10.1038/nclimate3377
Gutt, J., Arndt, J., Kraan, C., Dorschel, B., Schröder, M., Bracher, A., & Piepenburg, D. (2019). Benthic communities and their drivers: A spatial analysis off the Antarctic Peninsula. Limnology and Oceanography, 64(6), 2341–2357. https://doi.org/10.1002/lno.11187
Hajibabaei, M., Singer, G. A., Hebert, P. D., & Hickey, D. A. (2007). DNA barcoding: how it complements taxonomy, molecular phylogenetics and population genetics. Trends in Genetics, 23(4), 167–172. https://doi.org/10.1016/j.tig.2007.02.001
Handy, S. M., Deeds, J. R., Ivanova, N. V., Hebert, P. D., Hanner, R. H., & Yancy, H. F. (2011). A single-laboratory validated method for the generation of DNA barcodes for the identification of fish for regulatory compliance. Journal of AOAC International, 94(1), 201-210.
Hedgpeth, J. W. (1969). Preliminary Observations of Life Between Tidemarks at Palmer Station, 64 degrees 45, S 64 Degrees 05, W. Antarctic Journal of the United States, 4, 106.
Hillebrand, F. L., Bremer, U. F., de Freitas, M. W. D., Costi, J., Mendes Júnior, C. W., Arigony-Neto, J., & de Jesus, J. B. (2021). Statistical modeling of sea ice concentration in the northwest region of the Antarctic Peninsula. Environmental Monitoring and Assessment, 193(2), 1-22.
Hoffman, J. I., Peck, L. S., Hillyard, G., Zieritz, A., & Clark, M. S. (2010). No evidence for genetic differentiation between Antarctic limpet Nacella concinna morphotypes. Marine Biology, 157(4), 765–778. https://doi.org/10.1007/s00227-009-1360-5
Irisarri, I., Uribe, J. E., Eernisse, D. J., & Zardoya, R. (2020). A mitogenomic phylogeny of chitons (Mollusca: Polyplacophora). BMC Evolutionary Biology, 20(1), 1–15. https://doi.org/10.1186/s12862-019-1573-2
Irvine, G. V., & Shelly, A. (2013). Sampling design for long-term regional trends in marine rocky intertidal communities. Environmental Monitoring and Assessment, 185(8), 6963–6987.
Jossart, Q., Sands, C. J., & Sewell, M. A. (2019). Dwarf brooder versus giant broadcaster: combining genetic and reproductive data to unravel cryptic diversity in an Antarctic brittle star. Heredity, 123(5), 622–633. https://doi.org/10.1038/s41437-019-0228-9
Jossart, Q., Kochzius, M., Danis, B., Saucède, T., & Moreau, C. V. (2021). Diversity of the Pterasteridae (Asteroidea) in the Southern Ocean: a molecular and morphological approach. Zoological Journal of the Linnean Society, 192(1), 105–116. https://doi.org/10.1093/zoolinnean/zlaa097
Kearse, M., Moir, R., Wilson, A., Stones-Havas, S., & Drummond, A. (2012). Geneious Basic: an integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics, 28(12), 1647-1649. https://doi.org/10.1093/bioinformatics/bts199
Kelly, R. P., & Palumbi, S. R. (2010). Genetic structure among 50 species of the northeastern Pacific rocky intertidal community. PLoS One, 5(1), e8594. https://doi.org/10.1371/journal.pone.0008594
Kennicutt, M. C., Chown, S. L., Cassano, J. J., Liggett, D., Massom, R., & Sutherland, W. J. (2014). Polar research: six priorities for Antarctic science. Nature, 512(7512), 23-25. https://doi.org/10.1038/512023a
Kenny, R., & Haysom, N. (1962). Ecology of rocky shore organisms at Macquarie Island. Pacific Science, 16(3), 245–263.
Krishnamurthy, K. P., & Francis, R. A. (2012). A critical review on the utility of DNA barcoding in biodiversity conservation. Biodiversity and Conservation, 21(8), 1901–1919. https://doi.org/10.1007/s10531-012-0306-2
Krzeminska, M., & Kuklinski, P. (2018). Biodiversity patterns of rock encrusting fauna from the shallow sublittoral of the Admiralty Bay. Marine Environmental Research, 139, 169–181. https://doi.org/10.1016/j.marenvres.2018.03.016
Kuklinski, P., & Balazy, P. (2014). Scale of temperature variability in the maritime Antarctic intertidal zone. Journal of Sea Research, 85, 542–546. https://doi.org/10.1016/j.seares.2013.09.002
Laforest, B. J., Winegardner, A. K., Zaheer, O. A., Jeffery, N. W., Boyle, E. E., & Adamowicz, S. J. (2013). Insights into biodiversity sampling strategies for freshwater microinvertebrate faunas through bioblitz campaigns and DNA barcoding. BMC Ecology, 13(1), 1–17. https://doi.org/10.1186/1472-6785-13-13
Lagger, C., Servetto, N., Torre, L., & Sahade, R. (2017). Benthic colonization in newly ice-free soft-bottom areas in an Antarctic fjord. PLoS One, 12(11), e0186756. https://doi.org/10.1371/journal.pone.0186756
Layton, K.K., Corstorphine, E.A., & Hebert, P.D. (2016). Exploring Canadian echinoderm diversity through DNA barcodes. PLoS One, 11(11), e0166118. https://doi.org/10.1371/journal.pone.0166118
Lee, J. R., Raymond, B., Bracegirdle, T. J., Chades, I., Fuller, R. A., Shaw, J. D., & Terauds, A. (2017). Climate change drives expansion of Antarctic ice-free habitat. Nature, 547(7661), 49–54. https://doi.org/10.1038/nature22996
Legendre, L., & Legendre, P. (1983). Partitioning ordered variables into discrete states for discriminant analysis of ecological classifications. Canadian Journal of Zoology, 61(5), 1002–1010.
Leiva, C., Riesgo, A., Avila, C., Rouse, G. W., & Taboada, S. (2018). Population structure and phylogenetic relationships of a new shallow-water Antarctic phyllodocid annelid. Zoologica Scripta, 47(6), 714–726. https://doi.org/10.1111/zsc.12313
Levicoy, D., Flores, K., Rosenfeld, S., & Cárdenas, L. (2021). Phylogeography and genetic diversity of the microbivalve Kidderia subquadrata, reveals new data from West Antarctic Peninsula. Scientific Reports, 11(1), 1–12. https://doi.org/10.1038/s41598-021-85042-7
Levicoy, D., Rosenfeld, S., & Cárdenas, L. (2021). Divergence time and species delimitation of microbivalves in the Southern Ocean: the case of Kidderia species. Polar Biology, 44(7), 1365–1377. https://doi.org/10.1007/s00300-021-02885-6
López‐Farrán, Z., Guillaumot, C., Vargas‐Chacoff, L., Paschke, K., Dulière, V.., & Gérard, K. (2021). Is the southern crab Halicarcinus planatus (Fabricius, 1775) the next invader of Antarctica? Global Change Biology, 27(15), 3487-3504. https://doi.org/10.1111/gcb.15674
Mabragaña, E., Delpiani, S. M., Rosso, J. J., González-Castro, M., Deli Antoni, M., Hanner, R., & Díaz de Astarloa, J. M. (2016). Barcoding Antarctic fishes: species discrimination and contribution to elucidate ontogenetic changes in Nototheniidae. DNA Barcoding in Marine Perspectives, 213-242. https://doi.org/10.1007/978-3-319-41840-7_14
Maechler, M., Rousseeuw, P., Struyf, A., Hubert, M., Hornik, K. (2021). R package ‘cluster’. https://cran.r-project.org/web/packages/cluster/index.html
McCarthy, A. H., Peck, L. S., Hughes, K. A., & Aldridge, D. C. (2019). Antarctica: the final frontier for marine biological invasions. Global Change Biology, 25(7), 2221–2241. https://doi.org/10.1111/gcb.14600
McDermott, J. J., & Roe, P. (1985). Food, feeding behavior and feeding ecology of nemerteans. American Zoologist, 25(1), 113–125.
Meyer, C. P. (2003). Molecular systematics of cowries (Gastropoda: Cypraeidae) and diversification patterns in the tropics. Biological Journal of the Linnean Society, 79(3), 401–459. https://doi.org/10.1046/j.1095-8312.2003.00197.x
Molina, A. N., Pulgar, J. M., Rezende, E. L., & Carter, M. J. (2023). Heat tolerance of marine ectotherms in a warming Antarctica. Global Change Biology, 29(1), 179–188. https://doi.org/10.1111/gcb.16402
Moreau, C., Jossart, Q., Danis, B., Eléaume, M., Christiansen, H., Guillaumot, C., & Saucède, T. (2021). The high diversity of Southern Ocean sea stars (Asteroidea) reveals original evolutionary pathways. Progress in Oceanography, 190, 102472. https://doi.org/10.1016/j.pocean.2020.102472
Moreau, C., Saucede, T., Jossart, Q., Agüera, A., Brayard, A., & Danis, B. (2017). Reproductive strategy as a piece of the biogeographic puzzle: a case study using Antarctic sea stars (Echinodermata, Asteroidea). Journal of Biogeography, 44(4), 848–860. https://doi.org/10.1111/jbi.12965
Muths, D., Davoult, D., Gentil, F., & Jollivet, D. (2006). Incomplete cryptic speciation between intertidal and subtidal morphs of Acrocnida brachiata (Echinodermata: Ophiuroidea) in the Northeast Atlantic. Molecular Ecology, 15(11), 3303–3318. https://doi.org/10.1111/j.1365-294X.2006.03000.x
Oksanen, J., Blanchet, F. G., Friendly, M., Kindt, R., Legendre, P., & Wagner, H. (2020). R package ‘vegan’. https://github.com/vegandevs/vegan
O’Loughlin, P.M., & VandenSpiegel, D. (2010). A revision of Antarctic and some Indo-Pacific apodid sea cucumbers (Echinodermata: Holothuroidea: Apodida). Memoirs of Museum Victoria, 67(1).
Pante, E., Schoelinck, C., & Puillandre, N. (2015). From integrative taxonomy to species description: one step beyond. Systematic Biology, 64(1), 152–160. https://doi.org/10.1093/sysbio/syu083
Parry, G. D. (1981). The meanings of r- and K-selection. Oecologia, 48(2), 260–264.
Peck, L. S., Clark, M. S., & Dunn, N. I. (2018). Morphological variation in taxonomic characters of the Antarctic starfish Odontaster validus. Polar Biology, 41(10), 2159–2165. https://doi.org/10.1007/s00300-018-2344-z
Peck, L. S., Convey, P., & Barnes, D. K. (2006). Environmental constraints on life histories in Antarctic ecosystems: tempos, timings and predictability. Biological Reviews, 81(1), 75–109. https://doi.org/10.1017/S1464793105006871
Powell, A. W. B. (1951). Antarctic and subantarctic Mollusca: Pelecypoda and Gastropoda. Discovery Reports, 26, 49–196.
R Core Team. (2020). R: A language and environment for statistical computing. https://www.R-project.org/
Ratnasingham, S., & Hebert, P. D. (2007). BOLD: The Barcode of Life Data System (http://www.barcodinglife.org). Molecular Ecology Notes, 7(3), 355-364.
Robinson, B. J., Barnes, D. K., & Morley, S. A. (2020). Disturbance, dispersal and marine assemblage structure: A case study from the nearshore Southern Ocean. Marine Environmental Research, 160, 105025. https://doi.org/10.1016/j.marenvres.2020.105025
Rosenfeld, S., Maturana, C. S., Spencer, H. G., Convey, P., Saucède, T., Brickle, P., & Gonzalez-Wevar, C. (2022). Complete distribution of the genus Laevilitorina (Littorinimorpha: Littorinidae) in the Southern Hemisphere: remarks and natural history. ZooKeys, 1127, 61-77. https://doi.org/10.3897/zookeys.1127.91310
Saucède, T., Eléaume, M., Jossart, Q., Moreau, C., Downey, R., Bax, N., & Vignes-Lebbe, R. (2021). Taxonomy 2.0: computer-aided identification tools to assist Antarctic biologists in the field and in the laboratory. Antarctic Science, 33(1), 39-51. https://doi.org/10.1017/S0954102020000462
Schiaparelli, S., Danis, B., Wadley, V., & Stoddart, M. D. (2013). The census of Antarctic marine life: the first available baseline for Antarctic marine biodiversity. In Adaptation and Evolution in Marine Environments, 2, 3–19. https://doi.org/10.1007/978-3-642-27349-0_1
Schories, D., & Kohlberg, G. (Eds.). (2016). Marine Wildlife, King George Island (p. 348). Dirk Schories Publications.
Secretariat of the Antarctic Treaty. (2022). https://www.ats.aq/devAS/Ats/Guideline/39864605-7e82-4f8e-a171-49bdb8423e4f#
Siegert, M., Atkinson, A., Banwell, A., Brandon, M., Convey, P., & Vaughan, D. (2019). The Antarctic Peninsula under a 1.5°C global warming scenario. Frontiers in Environmental Science, 102. https://doi.org/10.3389/fenvs.2019.00102
Sluys, R., & De Vries, E. J. (1988). The aquatic triclads of the Crozet Islands (Platyhelminthes: Tricladida). Zoological journal of the Linnean Society, 94(3), 203–217. https://doi.org/10.1111/j.1096-3642.1988.tb01192.x
Stockton, W. L. (1973). An intertidal assemblage at Palmer Station. Antarctic Journal of the United States, 8(5), 305–307.
Sunnucks, P., & Hales, D. F. (1996). Numerous transposed sequences of mitochondrial cytochrome oxidase I-II in aphids of the genus Sitobion (Hemiptera: Aphididae). Molecular Biology and Evolution, 13(3), 510–524. https://doi.org/10.1093/oxfordjournals.molbev.a025612
Taboada, S., Junoy, J., Andrade, S., Giribet, G., Cristobo, J., & Avila, C. (2013). On the identity of two Antarctic brooding nemerteans: redescription of Antarctonemertes valida (Bürger, 1893) and description of a new species in the genus Antarctonemertes Friedrich, 1955 (Nemertea, Hoplonemertea). Polar Biology, 36(10), 1415–1430. https://doi.org/10.1007/s00300-013-1360-2
Tautz, D., Arctander, P., Minelli, A., Thomas, R. H., & Vogler, A. P. (2003). A plea for DNA taxonomy. Trends in Ecology & Evolution, 18(2), 70–74. https://doi.org/10.1016/S0169-5347(02)00041-1
Terauds, A., Chown, S. L., Morgan, F. J., Peat, H., Watts, D. J., & Bergstrom, D. M. (2012). Conservation biogeography of the Antarctic. Diversity and distributions, 18(7), 726–741. https://doi.org/10.1111/j.1472-4642.2012.00925.x
Teske, P. R., McQuaid, C. D., Froneman, P. W., & Barker, N. P. (2006). Impacts of marine biogeographic boundaries on phylogeographic patterns of three South African estuarine crustaceans. Marine Ecology Progress Series, 314, 283–293.
Thyrring, J., & Peck, L. S. (2021). Global gradients in intertidal species richness and functional groups. Elife, 10, e64541. https://doi.org/10.7554/eLife.64541
Valdivia, N., Díaz, M. J., Holtheuer, J., Garrido, I., Huovinen, P., & Gomez, I. (2014). Up, down, and all around: scale-dependent spatial variation in rocky-shore communities of Fildes Peninsula, King George Island, Antarctica. PLoS One, 9(6), e100714. https://doi.org/10.1371/journal.pone.0100714
Valdivia, N., Garrido, I., Bruning, P., Piñones, A., & Pardo, L. M. (2020). Biodiversity of an Antarctic rocky subtidal community and its relationship with glacier meltdown processes. Marine Environmental Research, 159, 104991. https://doi.org/10.1016/j.marenvres.2020.104991
Verberk, W. C. E. P. (2011). Explaining general patterns in species abundance and distributions. Nature Education Knowledge, 3(10), 38.
Waller, C. L. (2008). Variability in intertidal communities along a latitudinal gradient in the Southern Ocean. Polar Biology, 31(7), 809–816. https://doi.org/10.1007/s00300-008-0419-y
Waller, C. L. (2013). Zonation in a cryptic Antarctic intertidal macrofaunal community. Antarctic Science, 25(1), 62–68. https://doi.org/10.1017/S0954102012000867
Waller, C. L., Barnes, D. K., & Convey, P. (2006). Ecological contrasts across an Antarctic land–sea interface. Austral Ecology, 31(5), 656–666. https://doi.org/10.1111/j.1442-9993.2006.01618.x
Waller, C. L., Overall, A., Fitzcharles, E. M., & Griffiths, H. (2017). First report of Laternula elliptica in the Antarctic intertidal zone. Polar Biology, 40(1), 227–230. https://doi.org/10.1007/s00300-016-1941-y
Waller, C. L., Worland, M. R., Convey, P., & Barnes, D. K. A. (2006). Ecophysiological strategies of Antarctic intertidal invertebrates faced with freezing stress. Polar Biology, 29(12), 1077–1083. https://doi.org/10.1007/s00300-006-0152-3
Williams, S. T., Reid, D. G., & Littlewood, D. T. J. (2003). A molecular phylogeny of the Littorininae (Gastropoda: Littorinidae): unequal evolutionary rates, morphological parallelism, and biogeography of the Southern Ocean. Molecular Phylogenetics and Evolution, 28(1), 60–86. https://doi.org/10.1016/S1055-7903(03)00038-1
Yang, H. M., Ji, S. J., & Min, G. S. (2019). The complete mitochondrial genome of the Antarctic marine triclad, Obrimoposthia wandeli (Platyhelminthes, Tricladida, Maricola). Mitochondrial DNA Part B, 4(2), 2515–2516. https://doi.org/10.1080/2F23802359.2019.1640093
Zenteno, L., Cárdenas, L., Valdivia, N., Gómez, I., Höfer, J., Garrido, I., & Pardo, L. M. (2019). Unravelling the multiple bottom-up supplies of an Antarctic nearshore benthic community. Progress in Oceanography, 174, 55–63. https://doi.org/10.1016/j.pocean.2018.10.016
Acknowledgements
We thank Fernanda Azevedo, Antonio Carapelli, Gemma Collins, Charlène Guillaumot, Christoph Held, Bart Hellemans, Katrin Linse, Borja Mercado, Yi Ming Gan, Amy Moran, Lenka Nealova, Stefano Schiaparelli, Lucas Terrana, Anton Van de Putte, Ben Wallis and Helena Wiklund for their generous help and advice. We also thank the crew members (Ocean Expeditions) and scientists of the Belgica 121 expedition. Sebastian Rosenfeld would like to thank the Project ANID-Millennium Science Initiative Program - ICN2021_002.
Funding
This work was supported by the ‘Refugia and Ecosystem Tolerance in the Southern Ocean’ project (RECTO; BR/154/A1/RECTO) funded by the ‘Belgian Science Policy Office’ (BELSPO). This is contribution no. 28 to the RECTO project. The B121 expedition was funded by the Federation Wallonia-Brussels, the Fund for Scientific Research (FNRS), the Research Foundation – Flanders (FWO), the Leopold 3 Fund for the exploration and conservation of Nature and the Royal Belgian Society for Zoology. David Bauman was funded by the Belgian American Educational Foundation (BAEF) and the European Union's Horizon 2020 research and innovation programme under the Marie Skłodowska-Curie grant agreement No. 895799. Henrik Christiansen was funded by the BELSPO project COPE (B2/191/P1/COPE). Blanca Figuerola received funding from the Beatriu de Pinós program (2019-BP-00183), funded by the Secretary of Universities and Research (Government of Catalonia) and the European Union's Horizon 2020 program under the Marie Skłodowska-Curie grant agreement No. 801370. Peter Convey was supported by NERC core funding to the BAS ‘Biodiversity, Evolution and Adaptation’ Team.
Author information
Authors and Affiliations
Contributions
Conceived and designed the study: Quentin Jossart, Camille VE Moreau, Thomas Saucède, Bruno Danis; Performed fieldwork: Quentin Jossart, Camille VE Moreau, Thomas Saucède, Henrik Christiansen, Bruno Danis; Performed taxonomic work: Quentin Jossart, Camille VE Moreau, Thomas Saucède, Madeleine J Brasier, Peter Convey, Rachel Downey, Blanca Figuerola, Patrick Martin, Jon Norenburg, Sebastian Rosenfeld, Marie Verheye; Performed genetic laboratory work: Quentin Jossart, Henrik Christiansen; Analysed the data: Quentin Jossart, David Bauman, Bruno Danis; Wrote the paper: Quentin Jossart, David Bauman, Camille VE Moreau, Thomas Saucède, Henrik Christiansen, Madeleine J Brasier, Peter Convey, Rachel Downey, Blanca Figuerola, Patrick Martin, Jon Norenburg, Sebastian Rosenfeld, Marie Verheye, Bruno Danis.
Corresponding author
Ethics declarations
Ethical approval
All authors have read, understood, and have complied as applicable with the statement on "Ethical responsibilities of Authors" as found in the Instructions for Authors and are aware that with minor exceptions, no changes can be made to authorship once the paper is submitted.
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jossart, Q., Bauman, D., Moreau, C.V. et al. A pioneer morphological and genetic study of the intertidal fauna of the Gerlache Strait (Antarctic Peninsula). Environ Monit Assess 195, 514 (2023). https://doi.org/10.1007/s10661-023-11066-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10661-023-11066-3