Abstract
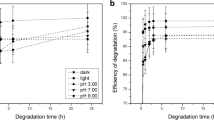
Organophosphate pesticides are used in large quantities. Once accumulated in the environment, they exhibit toxic effects on non-target organisms. Dimethoate, a frequently used insecticide, and its oxo-analog omethoate inhibit acetylcholinesterase and are toxic for mammals. However, under environmental conditions, they also undergo chemical transformations and decomposition. Nevertheless, the systematic data about dimethoate and omethoate decomposition are missing. We performed a systematic analysis of dimethoate and omethoate decomposition under different pH conditions and estimated their long-term eco-neurotoxic effects. Dimethoate and omethoate decompose rapidly in alkaline aqueous solutions (half-lives 5.7 ± 1.4 and 0.89 ± 0.21 days) but are highly stable in acidic solutions (half-lives 124 ± 18 and 104 ± 9 days). These differences are explained using quantum chemical calculations, indicating that a weaker P–S bond in omethoate is more susceptible to hydrolysis, particularly at a high pH. The toxicity of these pesticide solutions decreases over time, indicating that no or very little of highly more toxic omethoate is formed during hydrolysis of dimethoate, pointing to the advantage of alkaline hydrolysis over other techniques for dimethoate removal. Presented data are used to benchmark dimethoate and omethoate concentrations and toxicity in contaminated water in the pH range 3 to 9 for up to 70 days upon the release in the environment.
Graphical abstract







Similar content being viewed by others
Data Availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Acquaroni, M., Peluso, J., Svartz, G., Aronzon, C., & Perez, C. C. (2021). Characterization of acute toxicity, genotoxicity, and oxidative stress of dimethoate in Rhinella arenarum larvae. Environmental Science and Pollution Research International, 28(31), 41772–41779.
Baker, B. P., Benbrook, C. M., Iii, E. G., & Benbrook, K. L. (2002). Pesticide residues in conventional, integrated pest management (IPM)-grown and organic foods: Insights from three US data sets. Food Additives & Contaminants, 19(5), 427–446. https://doi.org/10.1080/02652030110113799
Blaudeau, J.-P., McGrath, M. P., Curtiss, L. A., & Radom, L. (1997). Extension of Gaussian-2 (G2) theory to molecules containing third-row atoms K and Ca. The Journal of Chemical Physics, 107(13), 5016–5021. https://doi.org/10.1063/1.474865
Clark, T., Chandrasekhar, J., Spitznagel, G. W., & Schleyer, P. V. R. (1983). Efficient diffuse function-augmented basis sets for anion calculations. III. The 3–21+G basis set for first-row elements Li-F. Journal of Computational Chemistry, 4(3), 294–301. https://doi.org/10.1002/jcc.540040303
Dai, P., Jack, C. J., Mortensen, A. N., Bustamante, T. A., Bloomquist, J. R., & Ellis, J. D. (2019). Chronic toxicity of clothianidin, imidacloprid, chlorpyrifos, and dimethoate to Apis mellifera L. larvae reared in vitro. Pest Management Science, 75(1), 29–36. https://doi.org/10.1002/ps.5124
Ellman, G. L., Courtney, K. D., Andres, V., J. R., & Featherstone, R. M. (1961). A new and rapid colorimetric determination of acetylcholinesterase activity. Biochemical Pharmacology, 7(2), 88–95. https://doi.org/10.1016/0006-2952(61)90145-9
Faust, S. D., & Gomaa, H. M. (1972). Chemical hydrolysis of some organic phosphorus and carbamate pesticides in aquatic environments. Environmental Letters, 3(3), 171–201. https://doi.org/10.1080/00139307209435465
Frisch, M. J., Pople, J. A., & Binkley, J. S. (1984). Self-consistent molecular orbital methods 25 Supplementary functions for Gaussian basis sets. The Journal of Chemical Physics, 80(7), 3265–3269. https://doi.org/10.1063/1.447079
Frisch M. J., Trucks G. W., Schlegel H. B., Scuseria G. E., M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, G. A. Petersson, H. Nakatsuji, X. Li M. C., A. Marenich, J. Bloino, B. G. Janesko, R. Gomperts, B. Mennucci, H. P. Hratchian, J. V. Ortiz, A. F. Izmaylov, J. L. Sonnenberg, D. Williams-Young, F. Ding, F. Lipparini, F. Egidi, J. Goings, B. Peng, A. Petrone, T. Henderson, D. Ranasinghe, V. G. Zakrzewski, J. Gao, N. Rega, G. Zheng, W. Liang, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, K. Throssell, J. A. Montgomery J., J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, T. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, J. M. Millam, M. Klene, C. Adamo, R. Cammi, J. W. Ochterski, R. L. Martin, K. Morokuma, O. Farkas, J. B. Foresman, Fox D. J. (2009) Gaussian 09, Revision d.01. Revision d.01 edn. Gaussian, Inc., Wallingford CT
https://www3.epa.gov/pesticides/chem_search/reg_actions/reregistration/ired_PC-035001_20-Aug-07.pdf
https://www.who.int/water_sanitation_health/WHS_WWD2010_guidelines_2010_6_en.pdf?ua=1
Hawker, D. (2015). Kinetics of carbaryl hydrolysis: An undergraduate environmental chemistry laboratory. Journal of Chemical Education, 92(9), 1531–1535. https://doi.org/10.1021/acs.jchemed.5b00097
Keith, T. A. (2019) AIMAll (Version 19.10.12). Version 19.10.12 edn. TK Gristmill Software, Overland Park KS, USA
Krishnan, R., Binkley, J. S., Seeger, R., & Pople, J. A. (1980). Self-consistent molecular orbital methods. XX. A basis set for correlated wave functions. The Journal of Chemical Physics, 72(1), 650–654. https://doi.org/10.1063/1.438955
Kunstadter, P., Prapamontol, T., Sirirojn, B.-O., Sontirat, A., Tansuhaj, A., & Khamboonruang, C. (2001). Pesticide exposures among hmong farmers in Thailand. International Journal of Occupational and Environmental Health, 7(4), 313–325. https://doi.org/10.1179/107735201800339227
Lazarevic-Pasti, T., Colovic, M., Savic, J., Momic, T., & Vasic, V. (2011). Oxidation of diazinon and malathion by myeloperoxidase. Pesticide Biochemistry and Physiology, 100(2), 140–144. https://doi.org/10.1016/j.pestbp.2011.03.001
Lazarevic-Pasti, T. D., Pasti, I. A., Jokic, B., Babic, B. M., & Vasic, V. M. (2016). Heteroatom-doped mesoporous carbons as efficient adsorbents for removal of dimethoate and omethoate from water. RSC Advances, 6(67), 62128–62139. https://doi.org/10.1039/c6ra06736k
Legradi, J. B., Di Paolo, C., Kraak, M. H. S., van der Geest, H. G., Schymanski, E. L., Williams, A. J., Dingemans, M. M. L., Massei, R., Brack, W., Cousin, X., Begout, M. L., van der Oost, R., Carion, A., Suarez-Ulloa, V., Silvestre, F., Escher, B. I., Engwall, M., NilénKeiter, G. S. H., Pollet, D., … Hollert, H. (2018). An ecotoxicological view on neurotoxicity assessment. Environmental Sciences Europe, 30(1), 46. https://doi.org/10.1186/s12302-018-0173-x
Lockridge, O., Verdier, L., & Schopfer, L. M. (2019). Half-life of chlorpyrifos oxon and other organophosphorus esters in aqueous solution. Chemico-Biological Interactions, 311, 108788. https://doi.org/10.1016/j.cbi.2019.108788
London, L., & de G. S., Wesseling C., Kisting S., Rother H. A., Mergler D. (2002). Pesticide usage and health consequences for women in developing countries: Out of sight, out of mind? International Journal of Occupational and Environmental Health, 8(1), 46–59. https://doi.org/10.1179/oeh.2002.8.1.46
McLean, A. D., & Chandler, G. S. (1980). Contracted Gaussian basis sets for molecular calculations I. Second row atoms, Z=11–18. The Journal of Chemical Physics, 72(10), 5639–5648. https://doi.org/10.1063/1.438980
Mitrović, T., Lazović, S., Nastasijević, B., Pašti, I. A., Vasić, V., & Lazarević-Pašti, T. (2019). Non-thermal plasma needle as an effective tool in dimethoate removal from water. Journal of Environmental Management, 246, 63–70. https://doi.org/10.1016/j.jenvman.2019.05.143
Mineau, P. (1991). Cholinesterase-inhibiting insecticides: Their impact on wildlife and the environment. Elsevier.
Pavlic, M., Haidekker, A., Grubwieser, P., & Rabl, W. (2002). Fatal intoxication with omethoate. International Journal of Legal Medicine, 116(4), 238–241.
Peshin, R., Hansra, B. S., Nanda, R., Singh, K., Sharma, R., Garg, L., Bajiya, M. R., Showkat, A., Kumar, R., & Yangsdon, S. (2020). Pesticides hazardous hotspots: Empirical evidences from North India. Environmental Management, 66(5), 899–915. https://doi.org/10.1007/s00267-020-01317-1
Rugno, G. R., Rugno, J. B. C., Qureshi, J. A., Stansly, P. A., & Yamamoto, P. T. (2021). Toxicity of imidacloprid, fenpropathrin, and dimethoate to Ceraeochrysa cubana (Neuroptera: Chrysopidae) through different exposure routes. Crop Protection., 146, 105649. https://doi.org/10.1016/j.cropro.2021.105649
Silva, V., Mol, H. G. J., Zomer, P., Tienstra, M., Ritsema, C. J., & Geissen, V. (2019). Pesticide residues in European agricultural soils - A hidden reality unfolded. Science of the Total Environment, 653, 1532–1545. https://doi.org/10.1016/j.scitotenv.2018.10.441
Tomasi, J., Mennucci, B., & Cammi, R. (2005). Quantum mechanical continuum solvation models. Chemical Reviews, 105(8), 2999–3093. https://doi.org/10.1021/cr9904009
Uhl, P., Franke, L. A., Rehberg, C., Wollmann, C., Stahlschmidt, P., Jeker, L., & Bruhl, C. A. (2016). Interspecific sensitivity of bees towards dimethoate and implications for environmental risk assessment. Scientific Reports, 6(1), 34439. https://doi.org/10.1038/srep34439
Van Scoy, A., Pennell, A., & Zhang, X. (2016). Environmental fate and toxicology of dimethoate. Reviews of Environmental Contamination and Toxicology, 237, 53–70. https://doi.org/10.1007/978-3-319-23573-8_3
Wang L., Hung Y., Lo H., Yapijakis C. (Eds.). (2004). Handbook of industrial and hazardous wastes treatment (2nd ed.). CRC Press. https://doi.org/10.1201/9780203026519
Wolfe, N. L., Zepp, R. G., Gordon, J. A., Baughman, G. L., & Cline, D. M. (1977). Kinetics of chemical degradation of malathion in water. Environment Science & Technology, 11(1), 88–93. https://doi.org/10.1021/es60124a001
Yan, X., Rong, R., Zhu, S., Guo, M., Gao, S., Wang, S., & Xu, X. (2015). Effects of ZnO nanoparticles on dimethoate-induced toxicity in mice. Journal of Agriculture and Food Chemistry, 63(37), 8292–8298.
Zhao, Y., & Truhlar, D. G. (2008). The M06 suite of density functionals for main group thermochemistry, thermochemical kinetics, noncovalent interactions, excited states, and transition elements: Two new functionals and systematic testing of four M06-class functionals and 12 other functionals. Theoretical Chemistry Accounts, 120(1), 215–241. https://doi.org/10.1007/s00214-007-0310-x
Funding
T.D.L.P. acknowledges the support provided by the Serbian Ministry of Education, Science and Technological Development (contract number: 451–03-68/2022–14/200017). M.P. and I.A.P. acknowledge the support provided by the Serbian Ministry of Education, Science and Technological Development (contract number: 451–03-68/2022–14/200146).
Author information
Authors and Affiliations
Contributions
Conceptualization: T.D.L.P. and I.A.P. Formal analysis: V.J.A., M.P., and T.D.L.P. Resources: M.P. and I.A.P. Writing—original draft preparation: all authors. Writing—review and editing: I.A.P., M.P., and T.D.L.P. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Anićijević, V.J., Petković, M., Pašti, I.A. et al. Decomposition of Dimethoate and Omethoate in Aqueous Solutions — Half-Life, Eco-Neurotoxicity Benchmarking, and Mechanism of Hydrolysis. Water Air Soil Pollut 233, 390 (2022). https://doi.org/10.1007/s11270-022-05861-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-022-05861-w