Abstract
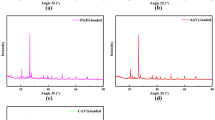
We investigated the structural changes in clay minerals after Cs adsorption and understood their low desorption efficiency using an ion-exchanger. We focused on the role of interlayers in Cs adsorption and desorption in 2:1 clay minerals, namely illite, hydrobiotite, and montmorillonite, using batch experiments and XRD and EXAFS analyses. The adsorption characteristics of the clay minerals were analyzed using cation exchange capacity (CEC), maximum adsorption isotherms (Qmax), and radiocesium interception potential (RIP) experiments. Although illite showed a low CEC value, it exhibited high selectivity for Cs with a relatively high RIP/CEC ratio. The Cs desorption efficiency after treatment with a NaCl ion exchanger was the highest for illite (74.3%), followed by hydrobiotite (45.5%) and montmorillonite (30.3%); thus, Cs adsorbed onto planar sites, rather than on interlayers or frayed edge sites (FESs), is easily desorbed. After NaCl treatment, XRD analysis showed that the low desorption efficiency was due to the collapse of the interlayer-fixed Cs, which tightly narrowed the interlayers’ hydrobiotite due to the ion exchange of divalent cations (Mg2+ or Ca2+) into the monovalent cation (Na+). Moreover, EXAFS analysis showed that hydrobiotite formed inner-sphere structures after NaCl desorption, indicating that it was difficult to remove Cs from NaCl desorption due to the collapsed hydrobiotite and montmorillonite interlayers as well as the strong bonding in FESs of illite. In contrast, chelation desorption using oxalic acid effectively dissolved the narrowed interlayers of hydrobiotite (98%) and montmorillonite (85.26%), enhancing the desorption efficiency. Therefore, low desorption efficiency for Cs clays using an ion exchanger was caused by the collapsed interlayer due to the exchange between monovalent cation and divalent cation.







Similar content being viewed by others
Data availability
Data and materials can be made available on request.
References
Abollino O, Giacomino A, Malandrino M, Mentasti E (2008) Interaction of metal ions with montmorillonite and vermiculite. Appl Clay Sci 38:227–236
Absalom J, Young S, Crout N, Nisbet A, Woodman R, Smolders E, Gillett A (1999) Predicting soil to plant transfer of radiocesium using soil characteristics. Environ Sci Technol 33:1218–1223
Akemoto Y, Sakti SCW, Kan M, Tanaka S (2021) Interpretation of the interaction between cesium ion and some clay minerals based on their structural features. Environ Sci Pollut Res 28:14121–14130
Bostick BC, Vairavamurthy MA, Karthikeyan K, Chorover J (2002) Cesium adsorption on clay minerals: an EXAFS spectroscopic investigation. Environ Sci Technol 36:2670–2676
Bourg IC, Sposito G, Bourg AC (2007) Modeling cation diffusion in compacted water-saturated sodium bentonite at low ionic strength. Environ Sci Technol 41:8118–8122
Bradbury MH, Baeyens B (2000) A generalised sorption model for the concentration dependent upta = ke of caesium by argillaceous rocks. J Contam Hydrol 42:141–163
Carroll D, Starkey HC (1971) Reactivity of clay minerals with acids and alkalies. Clays Clay Miner 19:321–333
Chen L, Dong Y (2013) Sorption of 63 Ni (II) to montmorillonite as a function of pH, ionic strength, foreign ions and humic substances. J Radioanal Nucl Chem 295:2117–2123
Cornell R (1993) Adsorption of cesium on minerals: a review. J Radioanal Nucl Chem 171:483–500
de Koning A, Comans RN (2004) Reversibility of radiocaesium sorption on illite. Geochim Cosmochim Acta 68:2815–2823
De Koning A, Konoplev A, Comans R (2007) Measuring the specific caesium sorption capacity of soils, sediments and clay minerals. Appl Geochem 22:219–229
Ding D, Zhang Z, Lei Z, Yang Y, Cai T (2016) Remediation of radiocesium-contaminated liquid waste, soil, and ash: a mini review since the Fukushima Daiichi Nuclear Power Plant accident. Environ Sci Pollut Res 23:2249–2263
Durrant CB, Begg JD, Kersting AB, Zavarin M (2018) Cesium sorption reversibility and kinetics on illite, montmorillonite, and kaolinite. Sci Total Environ 610:511–520
Dzene L, Tertre E, Hubert F, Ferrage E (2015) Nature of the sites involved in the process of cesium desorption from vermiculite. J Colloid Interface Sci 455:254–260
Fan Q, Xu J, Niu Z, Li P, Wu W (2012) Investigation of Cs (I) uptake on Beishan soil combined batch and EDS techniques. Appl Radiat Isot 70:13–19
Fan Q, Tanaka M, Tanaka K, Sakaguchi A, Takahashi Y (2014a) An EXAFS study on the effects of natural organic matter and the expandability of clay minerals on cesium adsorption and mobility. Geochim Cosmochim Acta 135:49–65
Fan Q, Yamaguchi N, Tanaka M, Tsukada H, Takahashi Y (2014b) Relationship between the adsorption species of cesium and radiocesium interception potential in soils and minerals: an EXAFS study. J Environ Radioact 138:92–100
Fu C, Tan Z, Cheng J, Xie J, Dai X, Du Y, Zhu S, Wang S, Yan M (2023) Effective removal of cesium by ammonium molybdophosphate–polyethylene glycol magnetic nanoparticles. J Environ Chem Eng 11:110544
Fukushi K, Sakai H, Itono T, Tamura A, Arai S (2014) Desorption of intrinsic cesium from smectite: inhibitive effects of clay particle organization on cesium desorption. Environ Sci Technol 48:10743–10749
Fuller AJ, Shaw S, Ward MB, Haigh SJ, Mosselmans JFW, Peacock CL, Stackhouse S, Dent AJ, Trivedi D, Burke IT (2015) Caesium incorporation and retention in illite interlayers. Appl Clay Sci 108:128–134
Han B, Zhang X, Liu C, Ma G, Guo D, Shao Y, Li P, Liang J, Fan Q (2023) Essential role of the interlayer of montmorillonite, vermiculite, and illite for Ni (II) sorption. J Radioanal Nucl Chem 332:1315–1323
Ho Y-S (2006) Review of second-order models for adsorption systems. J Hazard Mater 136:681–689
Hwang J, Han WS, Choung S, Kim J-W, Suk H, Lee J (2023) Diverse sorption capacities and contribution of multiple sorptive sites on illitic clays to assess the immobilization of dissolved cesium in subsurface environments. J Hazard Mater 441:129973
Iijima K, Tomura T, Shoji Y (2010) Reversibility and modeling of adsorption behavior of cesium ions on colloidal montmorillonite particles. Appl Clay Sci 49:262–268
Ikhsan J, Wells JD, Johnson BB, Angove MJ (2005) Surface complexation modeling of the sorption of Zn (II) by montmorillonite. Colloids Surf Physicochem Eng Aspects 252:33–41
Kikuchi R, Kogure T (2018) Structural and compositional variances in ‘hidrobiotite’ sample from Palabora, South Africa. Clay Science 22:39–52
Kim BH, Park CW, Yang H-M, Seo B-K, Lee B-S, Lee K-W, Park SJ (2017) Comparison of Cs desorption from hydrobiotite by cationic polyelectrolyte and cationic surfactant. Colloids Surf Physicochem Eng Aspects 522:382–388
Kim S-M, Yoon I-H, Kim I-G, Park CW, Sihn Y, Kim J-H, Park S-J (2020a) Cs desorption behavior during hydrothermal treatment of illite with oxalic acid. Environ Sci Pollut Res 27:35580–35590
Kim S-M, Yoon I-H, Kim I, Kim J-H, Park S-J (2020b) Hydrothermal desorption of Cs with oxalic acid from hydrobiotite and wastewater treatment by chemical precipitation. Energies 13:3284
Kim Y, Cygan RT, Kirkpatrick RJ (1996a) 133Cs NMR and XPS investigation of cesium adsorbed on clay minerals and related phases. Geochim Cosmochim Acta 60:1041–1052
Kim Y, Kirkpatrick RJ, Cygan RT (1996b) 133Cs NMR study of cesium on the surfaces of kaolinite and illite. Geochim Cosmochim Acta 60:4059–4074
Kogure T, Morimoto K, Tamura K, Sato H, Yamagishi A (2012) XRD and HRTEM evidence for fixation of cesium ions in vermiculite clay. Chem Lett 41:380–382
Komadel P, Madejová J, Janek M, Gates WP, Kirkpatrick R, Stucki JW (1996) Dissolution of hectorite in inorganic acids. Clays Clay Miner 44:228–236
Kumar KV, Ramamurthi V, Sivanesan S (2005) Modeling the mechanism involved during the sorption of methylene blue onto fly ash. J Colloid Interface Sci 284:14–21
Kwon S, Lim J, Seoung D, Cho Y, Park B (2023) Comparative study of the cesium adsorption behavior of montmorillonite and illite based on their mineralogical properties and interlayer cations. J Hazard Mater Adv 10:100258
Lalhriatpuia C, Tiwari D, Lee S-M (2014) Immobilized nickel hexacyanoferrate on activated carbons for efficient attenuation of radio toxic Cs (I) from aqueous solutions. Appl Surf Sci 321:275–282
Lee J, Park S-M, Jeon E-K, Baek K (2017) Selective and irreversible adsorption mechanism of cesium on illite. Appl Geochem 85:188–193
Liu X, Lu X, Wang R, Zhou H (2008) Effects of layer-charge distribution on the thermodynamic and microscopic properties of Cs-smectite. Geochim Cosmochim Acta 72:1837–1847
Miranda-Trevino JC, Coles CA (2003) Kaolinite properties, structure and influence of metal retention on pH. Appl Clay Sci 23:133–139
Motokawa R, Endo H, Yokoyama S, Nishitsuji S, Kobayashi T, Suzuki S, Yaita T (2014) Collective structural changes in vermiculite clay suspensions induced by cesium ions. Sci Rep 4:6585
Mukai H, Hatta T, Kitazawa H, Yamada H, Yaita T, Kogure T (2014) Speciation of radioactive soil particles in the Fukushima contaminated area by IP autoradiography and microanalyses. Environ Sci Technol 48:13053–13059
Nagy NM, Kónya J, Wazelischen-Kun G (1999) The adsorption and desorption of carrier-free radioactive isotopes on clay minerals and Hungarian soils. Colloids Surf Physicochem Eng Aspects 152:245–250
Okumura M, Nakamura H, Machida M (2013) Mechanism of strong affinity of clay minerals to radioactive cesium: first-principles calculation study for adsorption of cesium at frayed edge sites in muscovite. J Phys Soc Jpn 82:033802
Okumura T, Tamura K, Fujii E, Yamada H, Kogure T (2014) Direct observation of cesium at the interlayer region in phlogopite mica. Microscopy 63:65–72
Önal Y, Akmil-Başar C, Eren D, Sarıcı-Özdemir Ç, Depci T (2006) Adsorption kinetics of malachite green onto activated carbon prepared from Tunçbilek lignite. J Hazard Mater 128:150–157
Palansooriya KN, Yoon I-H, Kim S-M, Wang C-H, Kwon H, Lee S-H, Igalavithana AD, Mukhopadhyay R, Sarkar B, Ok YS (2022) Designer biochar with enhanced functionality for efficient removal of radioactive cesium and strontium from water. Environ Res 214:114072
Park CW, Kim I, Yoon I-H, Yang H-M, Seo B-K (2021a) Behaviors of desorption agents during removal of Cs from clay minerals and actual soil. J Nucl Fuel Cycle Waste Technol 19:39–49
Park CW, Kim S-M, Kim I, Yoon I-H, Hwang J, Kim J-H, Yang H-M, Seo BK (2021b) Sorption behavior of cesium on silt and clay soil fractions. J Environ Radioact 233:106592
Park S-M, Alessi DS, Baek K (2019a) Selective adsorption and irreversible fixation behavior of cesium onto 2: 1 layered clay mineral: a mini review. J Hazard Mater 369:569–576
Park S-M, Lee J, Jeon E-K, Kang S, Alam MS, Tsang DC, Alessi DS, Baek K (2019b) Adsorption characteristics of cesium on the clay minerals: structural change under wetting and drying condition. Geoderma 340:49–54
Poinssot C, Baeyens B, Bradbury MH (1999) Experimental and modelling studies of caesium sorption on illite. Geochim Cosmochim Acta 63:3217–3227
Qin H, Yokoyama Y, Fan Q, Iwatani H, Tanaka K, Sakaguchi A, Kanai Y, Zhu J, Onda Y, Takahashi Y (2012) Investigation of cesium adsorption on soil and sediment samples from Fukushima Prefecture by sequential extraction and EXAFS technique. Geochem J 46:297–302
Redlich O, Peterson DL (1959) A useful adsorption isotherm. J Phys Chem 63:1024–1024
Schnurr A, Marsac R, Rabung T, Lützenkirchen J, Geckeis H (2015) Sorption of Cm (III) and Eu (III) onto clay minerals under saline conditions: batch adsorption, laser-fluorescence spectroscopy and modeling. Geochim Cosmochim Acta 151:192–202
Tamura K, Kogure T, Watanabe Y, Nagai C, Yamada H (2014) Uptake of cesium and strontium ions by artificially altered phlogopite. Environ Sci Technol 48:5808–5815
Tan X, Fang M, Wang X (2010) Sorption speciation of lanthanides/actinides on minerals by TRLFS, EXAFS and DFT studies: a review. Molecules 15:8431–8468
Tournassat C, Tinnacher RM, Grangeon S, Davis JA (2018) Modeling uranium (VI) adsorption onto montmorillonite under varying carbonate concentrations: a surface complexation model accounting for the spillover effect on surface potential. Geochim Cosmochim Acta 220:291–308
Van Rompaey K, Van Ranst E, De Coninck F, Vindevogel N (2002) Dissolution characteristics of hectorite in inorganic acids. Appl Clay Sci 21:241–256
Wang W, Shi L, Wu H, Ding Z, Liang J, Li P, Fan Q (2023) Interactions between micaceous minerals weathering and cesium adsorption. Water Res 238:119918
Wu H, Qiang S, Fan Q, Zhao X, Liu P, Li P, Liang J, Wu W (2018) Exploring the relationship between Th (IV) adsorption and the structure alteration of phlogopite. Appl Clay Sci 152:295–302
Wu L, Liao L, Lv G (2015) Influence of interlayer cations on organic intercalation of montmorillonite. J Colloid Interface Sci 454:1–7
Yamamoto T (2012) Radioactivity of fission product and heavy nuclides deposited on soil in Fukushima Dai-Ichi Nuclear Power Plant accident: Fukushima NPP Accident Related. J Nucl Sci Technol 49:1116–1133
Yu S, Wang X, Chen Z, Tan X, Wang H, Hu J, Alsaedi A, Alharbi NS, Guo W, Wang X (2016) Interaction mechanism of radionickel on Na-montmorillonite: influences of pH, electrolyte cations, humic acid and temperature. Chem Eng J 302:77–85
Zachara JM, Smith SC, Liu C, McKinley JP, Serne RJ, Gassman PL (2002) Sorption of Cs+ to micaceous subsurface sediments from the Hanford site, USA. Geochim Cosmochim Acta 66:193–211
Zhang C, Liu X, Lu X, He M, Meijer EJ, Wang R (2017) Surface complexation of heavy metal cations on clay edges: insights from first principles molecular dynamics simulation of Ni (II). Geochim Cosmochim Acta 203:54–68
Zhang K, Li Z, Qi S, Chen W, Xie J, Wu H, Zhao H, Li D, Wang S (2022) Adsorption behavior of Cs (I) on natural soils: batch experiments and model-based quantification of different adsorption sites. Chemosphere 290:132636
Zhang K, Chen W, Lu M, Li Z, Qi S, Fan Q, Zhao H, Yang J, Li D, Zhang J (2023) Adsorption forms of cesium in specific soils based on EXAFS spectroscopic investigations and sequential extraction experiments. J Radioanal Nucl Chem 332:527–538
Zhang Y, Zhao H, Fan Q, Zheng X, Li P, Liu S, Wu W (2011) Sorption of U (VI) onto a decarbonated calcareous soil. J Radioanal Nucl Chem 288:395–404
Funding
This study was supported by grants from the National Research Foundation of Korea (NRF), funded by the Korean government, Ministry of Science, ICT and Future Planning (RS-2023–00243600 and 2022M2D2A1A02063569).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study’s conception and design. In-Ho Yoon is responsible for writing—original draft, conceptualization, methodology, software, data curation, visualization, and supervision. Sang-Ho Lee is assigned to the investigation, data curation, and writing—original draft. Ilgook Kim is responsible for the writing—review and editing, and validation. Sung Man Kim did the methodology, investigation, and data curation. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Tito Roberto Cadaval Jr
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yoon, IH., Lee, SH., Kim, I. et al. A comparative study on the Cs adsorption/desorption and structural changes in different clay minerals. Environ Sci Pollut Res 31, 25342–25355 (2024). https://doi.org/10.1007/s11356-024-32826-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-024-32826-9