Abstract
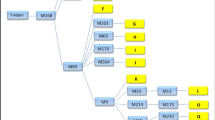
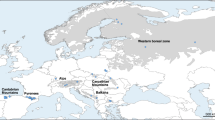
Drosophila bipectinata species complex includes four monophyletic closely related sympatric species with perfect reproductive isolation in natural conditions. Among these, two species, i.e., D. bipectinata and D. malerkotliana are known to be more prevalent in their occurrence all along their distribution area of Oriental–Australian regions. Genetic characteristics of these two species have been studied by earlier researchers mainly by focusing on their chromosomal polymorphism to gain knowledge pertaining to their evolutionary trend. The present work is one step farther, as we considered isozyme variants of three polymorphic enzymes to envisage genetic differentiation among the different natural populations of these two species, along with, a comparative perusal of their certain population genetic parameters. Natural populations of these two species were sampled from eight places aligned on the north–south axis of India and were subjected to isozyme analysis. The collection of both species and the geographical locations were kept same to avoid the effect of temporal or seasonal factors on the distribution of their allelic frequency. The results of this study clearly indicate that there is intraspecific genetic structuring among the populations of both the species with a distinct steady increase in the level of heterozygosity from north to south. The evolutionary tree in the form of dendrogram depicts two genetically differentiated groups of natural populations, one for north and other for South India. Most essential aspect that has been noticed through this study is the evolutionary trend of these two sympatric species being largely similar all along the north–south axis of India.









Similar content being viewed by others
References
Andersen DH, Pertoldi C, Loeschcke V, Cavicchi S, Scali V (2008) Divergence at neutral and non-neutral loci in Drosophila buzzatii populations and their hybrids. Evol Ecol 22(5):593–605
Arthur A, Weeks A, Sgro CM (2008) Investigating latitudinal clines for life history and stress resistance traits in Drosophila simulans from eastern Australia. J Evol Biol 21(6):1470–1479
Ayala FJ, Tracey ML, Barr LG, McDonald JF, Pérez-Salas S (1974) Genetic variation in natural populations of five Drosophila species and the hypothesis of the selective neutrality of protein polymorphisms. Genetics 77(2):343–384
Banerjee P, Singh BN (2012) Interspecific sexual isolation and phylogeny among different members of the Drosophila bipectinata species complex. Genetica 140(1):75–81
Banerjee P, Singh BN (2017) The Drosophila bipectinata species complex: phylogenetic relationship among different members based on chromosomal variations. J Genet 96(1):97–107
Barker J, Mulley J (1976) Isozyme variation in natural populations of Drosophila buzzatii. Evolution 30:213–233
Barker J, East P, Weir B (1986) Temporal and microgeographic variation in allozyme frequencies in a natural population of Drosophila buzzatii. Genetics 112(3):577–611
Bock I (1971) Taxonomy of the Drosophila bipectinata species complex. Stud Genet. 1971:273
Bock IR, Wheeler MR (1972) The Drosophila melanogaster species group. Tex Univ Publ 7213:1–102
Bubliy OA, Kalabushkin BA, Imasheva AG (1999) Geographic variation of six allozyme loci in Drosophila melanogaster: an analysis of data from different continents. Hereditas 130(1):25–32
Faleh AB, Shahin AA, Said K (2009) Allozyme polymorphism and genetic differentiation among populations of Jaculus jaculus and J. orientalis (Rodentia: Dipodidae) in Tunisia. Zool Res 3:247–254
Fry JD, Donlon K, Saweikis M (2008) A worldwide polymorphism in aldehyde dehydrogenase in Drosophila melanogaster: evidence for selection mediated by dietary ethanol. Evolution 62(1):66–75
Gupta J, Panigrahy K (1990) Chromosomal polymorphism in Indian populations of Drosophila bipectinata Duda. Genetica 82(1):45–49
Harris H (1966) C. Genetics of Man Enzyme polymorphisms in man. Proc R Soc Lond B Biol Sci. 164(995):298–310
Hegde S, Krishnamurthy N (1976) Studies on the genetics of isozymes in the hybrids of Drosophila bipectinata complex. Aust J Zool 27:421–431
Hoffmann A, Shirriffs J, Scott M (2005) Relative importance of plastic vs genetic factors in adaptive differentiation: geographical variation for stress resistance in Drosophila melanogaster from eastern Australia. Funct Ecol 19(2):222–227
Inoue Y, Tobari YN, Tsuno K, Watanabe TK (1984) Association of chromosome and enzyme polymorphisms in natural and cage populations of Drosophila melanogaster. Genetics 106(2):267–277
Jha AP, Mishra DN, Pandey BN (1979) Alcohol dehydrogenase isoenzymes in Drosophila species hybrids. Ind J Exp Biol 17:647–649
Kliman RM, Andolfatto P, Coyne JA, Depaulis F, Kreitman M, Berry AJ, McCarter J, Wakeley J, Hey J (2000) The population genetics of the origin and divergence of the Drosophila simulans complex species. Genetics 156(4):1913–1931
Kopp A, Barmina O (2005) Evolutionary history of the Drosophila bipectinata species complex. Genet Res 85(1):23–46
Krishnamoorti K, Singh AK (2017) Fitness differences due to allelic variation at esterase-4 locus in Drosophila ananassae. J Genet 96(4):625–631
Singh A, Kumar S, Bhumika (2013) Random genetic drift affecting Alcohol dehydrogenase polymorphism in laboratory populations of Drosophila ananassae. Jour Sci Res BHU 57:104–108
Kumar S, Singh A (2014) Latitudinal clines of allozymes in Indian natural populations of Drosophila ananassae. Dros Inf Serv 97:63–67
Kumar S, Singh A (2016) Allozyme polymorphism in Drosophila. Proc Zool Soc 1:22–31
Kumar S, Singh AK (2017) Population genetics of Drosophila: genetic variation and differentiation among Indian natural populations of Drosophila ananassae. Zool Stud 56:e1
Kumar S, Singh AK, Singh S (2019) Persistence of heterozygosity owing to balancing selection at allozyme loci in laboratory populations of Drosophila ananassae. Genomics Genet 12(1):11–18
Lewald KM, Abrieux A, Wilson DA, Lee Y, Conner WR, Andreazza F, Beers EH, Burrack HJ, Daane KM, Diepenbrock L (2021) Population genomics of Drosophila suzukii reveal longitudinal population structure and signals of migrations in and out of the continental United States. G3 11(12):jkab343
Lewontin RC, Hubby JL (1966) A molecular approach to the study of genic heterozygosity in natural populations. II. Amount of variation and degree of heterozygosity in natural populations of Drosophila pseudoobscura. Genetics 54(2):595
Mateus R, Sene F (2003) Temporal and spatial allozyme variation in the South American cactophilic Drosophila antonietae (Diptera; Drosophilidae). Biochem Genet 41(7):219–233
Mateus R, Sene F (2007) Population genetic study of allozyme variation in natural populations of Drosophila antonietae (Insecta, Diptera). J Zool Syst Evol Res 45(2):136–143
Mateus RP, Machado LP, Moraes EM, Sene FM (2010) Allozymatic divergence between border populations of two cryptic species of the Drosophila buzzatii cluster species (Diptera: Drosophilidae). Biochem Syst Ecol 38(3):410–415
Matsuda M, Tomimura Y, Tobari YN (2005) Reproductive isolation among geographical populations of Drosophila bipectinata Duda (Diptera, Drosophilidae) with recognition of three subspecies. Genetica 125(1):69–78
Matsuda M, Ng C-S, Doi M, Kopp A, Tobari Y (2009) Evolution in the Drosophila ananassae species subgroup. Fly 3(2):157–169
Morton RA, Choudhary M, Cariou M-L, Singh RS (2004) A reanalysis of protein polymorphism in Drosophila melanogaster, D. simulans, D. sechellia and D. mauritiana effects of population size and selection. Genetica 120(1):101–114
Nei M (1972) Genetic distance between populations. Am Nat 106:283–292
Ng CS, Hamilton AM, Frank A, Barmina O, Kopp A (2008) Genetic basis of sex-specific color pattern variation in Drosophila malerkotliana. Genetics 180(1):421–429
Oakeshott J, Gibson J, Anderson P, Knibb W, Anderson D, Chambers G (1982) Alcohol dehydrogenase and glycerol-3-phosphate dehydrogenase clines in Drosophila melanogaster on different continents. Evolution 36:86–96
Ometto L, Glinka S, De Lorenzo D, Stephan W (2005) Inferring the effects of demography and selection on Drosophila melanogaster populations from a chromosome-wide scan of DNA variation. Mol Biol Evol 22(10):2119–2130
Parkash R, Singh D, Lambhod C (2014) Divergent strategies for adaptations to stress resistance in two tropical Drosophila species: effects of developmental acclimation in D. bipectinata and the invasive species D. malerkotliana. J Exp Biol 217(6):924–934
Peakall R, Smouse PE (2012) GenAlEx 6,5, genetic analysis in Excel: Population genetic software for teaching and research, an update. Bioinformatics 28:2537–2539
Pecsenye K, Saura A (2002) Structure of variation in enzyme activity in natural Drosophila melanogaster populations. Hereditas 136(1):75–83
Petermann S, Otto S, Eichner G, Schetelig MF (2021) Spatial and temporal genetic variation of Drosophila suzukii in Germany. J Pest Sci 94:1–15
Prakash S (1977) Allelic variants at the xanthine dehydrogenase locus affecting enzyme activity in Drosophila pseudoobscura. Genetics 87(1):159–168
Sharma M, Sharma S, Parkash R (1993) ADH polymorphism and ethanol tolerance in three species of ananassae species subgroup. Evol Biol 7:51–62
Singh BN (2013) Genetic polymorphisms in Drosophila. Curr Sci 105:461–469
Singh BN, Banerjee P (2016) Population genetical, behavioural and evolutionary studies in the Drosophila bipectinata species complex. Proc Indian Nat Sci Acad 82:99–115
Singh G, Singh A (2018) Excessive occurrence of paracentric inversions in a Natural population of Drosophila bipectinata. J Exp Zool India 21:29–33
Singh G, Singh A (2020) Non-random distribution of heterozygous inversions in a natural population of Drosophila malerkotliana. J Sci Res 64(1):85
Singh G, Singh AK (2021) Genetic structuring of Drosophila bipectinata in Indian natural populations based on the distribution of cosmopolitan inversions. J Genet 100(2):1–10
Singh AK, Yadav N, Singh G (2017) Isozyme variants in two natural populations of Lymnaea luteola. Eur J Biol Res 7(4):360–365
Tomimura Y, Matsuda M, Tobari YN (2005) Chromosomal phylogeny and geographical divergence in the Drosophila bipectinata complex. Genome 48(3):487–502
Umina PA, Weeks AR, Kearney MR, McKechnie SW, Hoffmann AA (2005) A rapid shift in a classic clinal pattern in Drosophila reflecting climate change. Science 308(5722):691–693
Voelkar RA, Langley CH, Leigh-Brown AJ, Ohnishi S, Kickson B, Montgomeri E, Smith SC (1980) Enzyme null alleles in natural populations of Drosophila melanogaster. Proc Natl Acad Sci USA 77:1091–1095
Wright S (1943) Isolation by distance. Genetics 28:114
Yang S (1972) Isozyme variations and phylogenetic relationships in the Drosophila bipectinata species complex. Univ Texas Publ 7213:213–227
Yue L, Cao LJ, Chen JC et al (2021) Low levels of genetic differentiation with isolation by geography and environment in populations of Drosophila melanogaster from across China. Heredity. https://doi.org/10.1038/s41437-021-00419-8
Acknowledgements
We sincerely thank the anonymous reviewers for their valuable comments and suggestions on the manuscript for its improvement. Authors are also thankful to University Grants Commission and Indian Council of Medical Research (ICMR), New Delhi for providing research fellowship in the form of SRF (2019-4830/GEN-BMS) to GS. We earnestly thank the unspecified persons who met at the flies’ collection sites as a matter of chance and helped in their possible capacity.
Funding
Indian Council of Medical Research (ICMR), New Delhi is highly acknowledged for providing financial support (Project code- 2019–4830/GEN-BMS).
Author information
Authors and Affiliations
Contributions
GS conducted experiments, collected and analyzed the data. AKS conceived and designed the research. GS drafted the manuscript. AKS revised the final version. Both the authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they do not have any competing interests.
Rights and permissions
About this article
Cite this article
Singh, G., Singh, A.K. Intra- and interspecific comparison of protein polymorphism to establish genetic differentiation in two sympatric species of Drosophila: D. bipectinata and D. malerkotliana. 3 Biotech 12, 195 (2022). https://doi.org/10.1007/s13205-022-03257-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-022-03257-5