Abstract
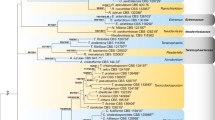
Caves are underground and natural environments mainly found in rocky terrain. Caves have a very specific microclimate, which benefits the occurrence of specific fungi. In recent studies, researchers have observed that caves harbour a great diversity of fungi. However, studies on fungal diversity in Brazilian caves are still incipient. In September 2019, airborne spore and soil samples were collected from the Monte Cristo cave, in the Southern Espinhaço Range, Diamantina, Minas Gerais state, Brazil. Two Cladosporium single‐spore isolates, among other genera, were obtained from these samples. This study aimed to characterise these two fungal isolates based on their DNA sequence data and morphology. Phylogenetic analyses of the rDNA-ITS, ACT and TEF1-α loci revealed that the isolates belonged to the Cladosporium cladosporioides species complex. Both isolates did not cluster with any known species and were formally described and named herein as C. diamantinense and C. speluncae. This study presents taxonomic novelties and contributes to the knowledge about the fungal diversity in Brazilian caves.




Similar content being viewed by others
Data availability
DNA sequences generated in this study are available in GenBank (https://www.ncbi.nlm.nih.gov/genbank).
References
Poulson TL, White WB (1969) The cave environment. Science 165:971–981. https://doi.org/10.1126/science.165.3897.971
Fernandez-Cortes A, Cuezva S, Sanchez-Moral S, Carlos Cañaveras J, Porca E, Jurado V, Martin-Sanchez PM, Saiz-Jimenez C (2011) Detection of human-induced environmental disturbances in a show cave. Environ Sci Pollut Res 18:1037–1045. https://doi.org/10.1007/s11356-011-0513-5
Kokurewicz T, Ogórek R, Pusz W, Matkowski K (2016) Bats increase the number of cultivable airborne fungi in the “Nietoperek” bat reserve in Western Poland. Microb Ecol 72:36–48. https://doi.org/10.1007/s00248-016-0763-3
Zhang ZF, Liu F, Zhou X, Liu XZ, Liu SJ, Cai L (2017) Culturable mycobiota from Karst caves in China, with descriptions of 20 new species. Persoonia 39:1–31. https://doi.org/10.3767/persoonia.2017.39.01
Zhang ZF, Zhou S-Y, Eurwilaichitr L, Ingsriswang S, Raza M et al (2020) Culturable mycobiota from Karst caves in China II, with descriptions of 33 new species. Fungal Divers 106:29–136. https://doi.org/10.1007/s13225-020-00453-7
Vanderwolf KJ, Malloch D, Mcalpine DF, Forbes GJ (2013) A world review of fungi, yeasts, and slime molds in caves. Int J Speleol 42:77–96. https://doi.org/10.5038/1827-806X.42.1.9
Cunha AOB, Bezerra JDP, Oliveira TGL, Barbier E, Bernard E, Machado AR, Souza-Motta CM (2020) Living in the dark: bat caves as hotspots of fungal diversity. PLoS ONE 15:e0243494. https://doi.org/10.1371/journal.pone.0243494
Pereira MLS, Carvalho JLVR, Lima JMS, Barbier E, Bernard E, Bezerra JDP, Souza-Motta CM (2022) Richness of Cladosporium in a tropical bat cave with the description of two new species. Mycol Prog 21:345–357. https://doi.org/10.1007/s11557-021-01760-2
Carvalho JLVR, Lima JMS, Barbier E et al (2022) Ticket to ride: fungi from bat ectoparasites in a tropical cave and the description of two new species. Braz J Microbiol 53:2077–2091. https://doi.org/10.1007/s42770-022-00841-y
Alves VCS, Lira RA, Lima JMS et al (2022) Unravelling the fungal darkness in a tropical cave: richness and the description of one new genus and six new species. Fungal Syst Evol 10:139–167. https://doi.org/10.3114/fuse.2022.10.06
Taylor ELS, Resende-Stoianoff MA, Lopes Ferreira R (2013) Mycological study for a management plan of a neotropical show cave (Brazil). Int J Speleol 42:267–277. https://doi.org/10.5038/1827-806X.42.3.10
Taylor ELS, Ferreira RL, Cardoso PG et al (2014) Cave entrance dependent spore dispersion of filamentous fungi isolated from various sediments of iron ore cave in Brazil: a colloquy on human threats while caving. Ambient Science 1:16–28. https://doi.org/10.21276/ambi.2014.01.1.ra02
Abdollahzadeh J, Groenewald JZ, Coetzee MPA, Wingfeld MJ, Crous PW (2020) Evolution of lifestyles in Capnodiales. Stud Mycol 95:381–414. https://doi.org/10.1016/j.simyco.2020.02.004
Bensch K, Braun U, Groenewald JZ, Crous PW (2012) The genus Cladosporium. Stud Mycol 72:1–401. https://doi.org/10.3114/sim0003
David JC (1997) A contribution to the systematics of Cladosporium. Revision of the fungi previously referred to Heterosporium. Mycol Pap 172:1–157
Braun U, Crous PW, Dugan F, Groenewald JZE, de Hoog GS (2003) Phylogeny and taxonomy of Cladosporium-like hyphomycetes, including Davidiella gen. nov., the teleomorph of Cladosporium s. str. Mycol Prog 2:3–18. https://doi.org/10.1007/s11557-006-0039-2
Crous PW, Braun U, Schubert K, Groenewald JZ (2007) Delimiting Cladosporium from morphologically similar genera. Stud Mycol 58:33–56. https://doi.org/10.3114/sim.2007.58.02
Bensch K, Groenewald JZ, Meijer M, Dijksterhuis J, Jurjevic Z, Andersen B, Houbraken J, Crous PW, Samson RA (2018) Cladosporium species in indoor environments. Stud Mycol 89:177–301. https://doi.org/10.1016/j.simyco.2018.03.002
Schubert K, Groenewald JZ, Braun U, Dijksterhuis J, Starink M, Hill CF, Zalar P, de Hoog GS, Crous PW (2007) Biodiversity in the Cladosporium herbarum complex (Davidiellaceae, Capnodiales), with standardisation of methods for Cladosporium taxonomy and diagnostics. Stud Mycol 58:105–156. https://doi.org/10.3114/sim.2007.58.05
Bensch K, Groenewald JZ, Dijksterhuis J, Starink-Willemse M, Andersen B et al (2010) Species and ecological diversity within the Cladosporium cladosporioides complex (Davidiellaceae, Capnodiales). Stud Mycol 67:1–94. https://doi.org/10.3114/sim.2010.67.01
Dugan FM, Braun U, Groenewald JZ, Crous PW (2008) Morphological plasticity in Cladosporium sphaerospermum. Persoonia 21:9–16. https://doi.org/10.3767/003158508X334389
Ma R, Chen Q, Fan Y, Wang Q, Chen S et al (2017) Six new soil-inhabiting Cladosporium species from plateaus in China. Mycologia 109:244–260. https://doi.org/10.1080/00275514.2017.1302254
Crous PW, Wingfield MJ, Burgess TI, Hardy GESTJ, Gené J et al (2018) Fungal Planet description sheets: 716–784. Persoonia 40:240–393. https://doi.org/10.3767/persoonia.2018.40.10
Bensch K, Groenewald JZ, Braun U, Dijksterhuis J, MdeJ Y-M, Crous PW (2015) Common but different: the expanding realm of Cladosporium. Stud Mycol 82:23–74. https://doi.org/10.1016/j.simyco.2015.10.001
Abdel-Baky NF, Abdel-Salam AH (2003) Natural incidence of Cladosporium spp. as a bio-control agent against whiteflies and aphids in Egypt. J Appl Entomol 127:228–235. https://doi.org/10.1046/j.1439-0418.2003.00662.x
Köhl J, Scheer C, Holb IJ, Masny S, Molhoek WML (2015) Toward an integrated use of biological control by Cladosporium cladosporioides H39 in apple scab (Venturia inaequalis) management. Plant Dis 99:535–543. https://doi.org/10.1094/PDIS-08-14-0836-RE
Torres DE, Rojasmartínez RI, Zavaletamejía E, Guevarafefer P, Márquezguzmán GJ, Pérezmartínez C (2017) Cladosporium cladosporioides and Cladosporium pseudocladosporioides as potential new fungal antagonists of Puccinia horiana Henn., the causal agent of chrysanthemum white rust. PLoS ONE 12:e0170782. https://doi.org/10.1371/journal.pone.0170782
Zalar P, de Hoog GS, Schroers H-J, Crous PW, Groenewald JZ, Gunde-Cimerman N (2007) Phylogeny and ecology of the ubiquitous saprobe Cladosporium sphaerospermum, with descriptions of seven new species from hypersaline environments. Stud Mycol 58:157–183. https://doi.org/10.3114/sim.2007.58.06
Grum-Grzhimaylo AA, Georgieva ML, Bondarenko SA, Debets AJM, Bilanenko EN (2016) On the diversity of fungi from soda soils. Fungal Divers 76:27–74. https://doi.org/10.1007/s13225-015-0320-2
Porca E, Jurado V, Martin-Sanchez PM, Hermosin B, Bastian F et al (2011) Aerobiology: an ecological indicator for early detection and control of fungal outbreaks in caves. Ecol Indic 11:1594–1598. https://doi.org/10.1016/j.ecolind.2011.04.003
Ogórek R, Lejman A, Matkowski K (2014) Influence of external environment on airborn fungi isolated from a cave. Pol J Environ Stud 23:435–440
Sandoval-Denis M, Gené J, Sutton DA, Wiederhold NP, Cano-Lira JF, Guarro T (2016) New species of Cladosporium associated with human and animal infections. Persoonia 36:281–298. https://doi.org/10.3767/003158516X691951
Hawksworth DL (2001) The magnitude of fungal diversity: the 1.5 million species estimate revisited. Mycol Res 105:1422–1432. https://doi.org/10.1017/S0953756201004725
Senanayake IC, Pem D, Rathnayaka AR, Wijesinghe SN, Tibpromma S et al (2022) Predicting global numbers of teleomorphic ascomycetes. Fungal Divers 114:237–278. https://doi.org/10.1007/s13225-022-00498-w
Verdi M, Pougy N, Martins E, Martinelli G (2015) A Serra do Espinhaço Meridional. In: Plano de ação nacional para a conservação da fora ameaçada de extinção da Serra do Espinhaço Meridional. CNCFlora:Jardim Botânico do Rio de Janeiro:Laboratório de Biogeografa da Conservação, Rio de Janeiro, pp 19–32. https://dspace.jbrj.gov.br/jspui/bitstream/doc/42/1/PAN_Serra%20Espinhaco%20Meridional.pdf. Accessed 21 Sept 2023
Almeida-Abreu PA, Pflug R (1994) The geodynamic evolution of the southern Serra do Espinhaço, Minas Gerais, Brazil. Zent bl Geol Paläontologie Teil I:21–44
Braga PLM, Sá FS, Gradanucci JPL (2011) Ecologia Populacional de Aranhas Troglófilas Trechona sp. (Mygalomorphae, Dipluridae) na Gruta Monte Cristo, Diamantina, Minas Gerais. In: Congresso Brasileiro de Espeleologia, 31, Ponta Grossa. Anais. SBE, Ponta Grossa, pp 501–505.
Guadanucci JPL, Braga PLM, FdeS Sá (2015) Aspects of the activity rhythm and population size of troglophilic mygalomorph spiders (Trechona sp., Dipluridae) in a quartzite cave in Minas Gerais. Brazil J Nat Hist 49:889–903. https://doi.org/10.1080/00222933.2014.946108
Borda D, Borda C, Tămaş T (2004) Bats, climate, and air microorganisms in a Romanian cave. Mammalia 68(4):337–343. https://doi.org/10.1515/mamm.2004.033
Kuzmina LY, Galimzianova NF, Abdullin SR, Ryabova AS (2012) Microbiota of the Kinderlinskaya cave (South Urals, Russia). Microbiol 81:251–258. https://doi.org/10.1134/S0026261712010109
Dhingra OD, Sinclair JB (1995) Slide culture techniques and preparation of permanent mounts. In: Dhingra OD, Sinclair JB (eds) Basic plant pathology methods, 2nd edn. CRC Press, Boca Raton, Florida, pp 319–320
Gonçalves RC, Alfenas AC, Mafia RG (2016) Armazenamento de microrganismos em cultura com ênfase em fungos fitopatogênicos. In: Alfenas AC, Mafia RG (eds) Métodos em Fitopatologia, 2nd edn. Editora UFV, Viçosa, pp 93–105
Pinho DB, Firmino AL, Ferreira-Junior WG, Pereira OL (2012) An eficiente protocol for DNA extraction from Meliolales and the description of Meliola centellae sp. nov. Mycotaxon 122:333–345. https://doi.org/10.5248/122.333
White TJ, Bruns T, Lee S, Taylor J (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ. (eds) PCR Protocols: a guide to methods and applications. Academic Press, San Diego, California, USA, pp 315–322. https://doi.org/10.1016/B978-0-12-372180-8.50042-1
Carbone I, Kohn LM (1999) A method for designing primer sets for speciation studies in filamentous Ascomycetes. Mycologia 91:553–556. https://doi.org/10.1080/00275514.1999.12061051
Rosado AWC, Custódio FA, Pinho DB, Ferreira APS, Pereira OL (2019) Cladosporium species associated with disease symptoms on Passiflora edulis and other crops in Brazil, with descriptions of two new species. Phytotaxa 409:239–260. https://doi.org/10.11646/phytotaxa.409.5.1
Katoh K, Standley DM (2013) MAFFT Multiple Sequence Alignment Software version 7: improvements in performance and usability. Mol Biol Evol 30:772–780. https://doi.org/10.1093/molbev/mst010
Kumar S, Stecher G, Tamura K (2016) MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol 33:1870–1874. https://doi.org/10.1093/molbev/msw054
Posada D, Buckley TR (2004) Model selection and model averaging in phylogenetics: advantages of Akaike Information Criterion and Bayesian approaches over likelihood ratio tests. Syst Biol 53:793–808. https://doi.org/10.1080/10635150490522304
Miller MA, Schwartz T, Pickett BE, He S, Klem EB, Scheuermann RH, Passarotti M, Kaufman S, O’Leary MA (2015) A RESTful API for access to phylogenetic tools via the CIPRES Science Gateway. Evol Bioinform Online 11:43–48. https://doi.org/10.4137/EBO.S21501
Ronquist F, Teslenko M, Van Der Mark P, Ayres DL, Darling A, Höhna S, Larget B, Liu L, Suchard MA, Huelsenbeck JP (2012) MrBayes 3.2: efficient Bayesian phylogenetic inference and model choice across a large model space. Syst Biol 61:539–542. https://doi.org/10.1093/sysbio/sys029
Rannala BH, Yang Z (1996) Probability distribution of molecular evolutionary trees: a new method of phylogenetic inference. J Mol Evol 43:304–311. https://doi.org/10.1007/BF02338839
Nguyen L-T, Schmidt HA, von Haeseler A, Minh BQ (2015) IQ-TREE: a fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol Biol Evol 32:268–74. https://doi.org/10.1093/molbev/msu300
Hoang DT, Chernomor O, von Haeseler A, Minh BQ, Vinh LS (2018) UFBoot2: improving the ultrafast bootstrap approximation. Mol Biol Evol 35:518–22. https://academic.oup.com/mbe/article/35/2/518/4565479. Accessed 11 Jun 2022
Rambaut A (2018) FigTree – tree figure drawing tool, v1.4.4. Molecular evolution, phylogenetics and epidemiology. http://tree.bio.ed.ac.uk/software/figtree/. Accessed 11 Jun 2022
Rayner RW (1970) A mycological colour chart. CMI and British Mycological Society, Kew, Surrey, England, UK
Schoch CL, Seifert KA, Huhndorf S, Robert V, Spouge JL et al (2012) Nuclear ribosomal internal transcribed spacer (ITS) region as a universal DNA barcode marker for Fungi. Proc Natl Acad Sci USA 109:6241–6246. https://doi.org/10.1073/pnas.1117018109
Marin-Felix Y, Groenewald JZ, Cai L, Chen Q, Marincowitz S (2017) Genera of phytopathogenic fungi: GOPHY 1. Stud Mycol 86:99–216. https://doi.org/10.1016/j.simyco.2017.04.002
Yang Y, Luo W, Zhang W, Mridha MAU, Wijesinghe SN, McKenzie EHC, Wang Y (2023) Cladosporium species associated with fruit trees in Guizhou province, China. J Fungi 9(2):250. https://doi.org/10.3390/jof9020250
Crous PW, Osieck ER, Shivas RG et al (2023) Fungal Planet description sheets: 1478–1549. Persoonia 50:220–221. https://doi.org/10.3767/persoonia.2023.50.05
Lee W, Kim JS, Seo CW, Lee JW, Kim SH, Cho Y, Lim YW (2023) Diversity of Cladosporium (Cladosporiales, Cladosporiaceae) species in marine environments and report on five new species. MycoKeys 98:87–111. https://doi.org/10.3897/mycokeys.98.101918
Viégas AP (1946) Alguns Fungos do Brasil XIII - Hifomicetos. Bragantia 6(8):367–368
Freitas MLR, Gomes AAM, Rosado AWC, Pereira OL (2021) Cladosporium species from submerged decayed leaves in Brazil, including a new species and new records. Phytotaxa 482:223–239. https://doi.org/10.11646/phytotaxa.482.3.1
Costa PP, Rosado AWC, Pereira OL (2022) Six new species of Cladosporium associated with decayed leaves of native bamboo (Bambusoideae) in a fragment of Brazilian Atlantic Forest. Phytotaxa 560:001–029. https://doi.org/10.11646/phytotaxa.560.1.1
Acknowledgements
We thank colleagues of the Laboratório de Micologia e Etiologia de Doenças Fúngicas de Plantas/UFV for their collaboration during the conception of this work.
Funding
This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior, Brasil (CAPES, Finance Code 001), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and Fundação de Amparo à Pesquisa do Estado de Minas Gerais (FAPEMIG-VALE RDP-00017–18).
Author information
Authors and Affiliations
Contributions
A. W. C. R., A. F. L., T. O. C. and O. L. P. collected the samples. S. d. C. N. and L. M. S. F. carried out the caving and sampling. Y. L. G. D. processed the samples, carried out the fungal isolations, extracted DNA and did amplifications with the help of A. F. L., A. W. C. R. and T. O. C. T. O. C., A. W. C. R., Y. L. G. D. and O. L. P carried out the phylogenetic analyses and data interpretation. M. C. M. K. and O. L. P. conceived and designed the research project. A. W. C. R. and Y. L. G. D. wrote the manuscript with input from all authors.
Corresponding author
Ethics declarations
Ethics approval
The collection was authorised by the Ministério do Meio Ambiente (MMA)/Instituto Chico Mendes de Conservação da Biodiversidade (ICMBio) (SISBIO number 70978–1).
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Responsible Editor: Melissa Fontes Landell
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Dutra, Y.L.G., Rosado, A.W.C., Condé, T.O. et al. Two new Cladosporium species from a quartzite cave in Brazil. Braz J Microbiol 54, 3021–3031 (2023). https://doi.org/10.1007/s42770-023-01156-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42770-023-01156-2