💊 𝐔𝐭𝐢𝐥𝐢𝐳𝐚𝐭𝐢𝐨𝐧 𝐨𝐟 𝐏𝐚𝐫𝐤𝐢𝐧𝐬𝐨𝐧’𝐬 𝐝𝐢𝐬𝐞𝐚𝐬𝐞 𝐩𝐚𝐭𝐢𝐞𝐧𝐭 𝐬𝐩𝐞𝐜𝐢𝐟𝐢𝐜 𝐬𝐚𝐦𝐩𝐥𝐞𝐬 𝐟𝐨𝐫 𝐭𝐡𝐞𝐫𝐚𝐩𝐞𝐮𝐭𝐢𝐜𝐬 𝐝𝐢𝐬𝐜𝐨𝐯𝐞𝐫𝐲 – 𝐉𝐮𝐧𝐭𝐞𝐧𝐝𝐨 𝐔𝐧𝐢𝐯𝐞𝐫𝐬𝐢𝐭𝐲 𝐚𝐧𝐝 𝐎𝐫𝐠𝐚𝐧𝐨𝐓𝐡𝐞𝐫𝐚𝐩𝐞𝐮𝐭𝐢𝐜𝐬 𝐰𝐨𝐫𝐤 𝐭𝐨𝐠𝐞𝐭𝐡𝐞𝐫 🤝 Patient specific stem cell lines are the foundation for the development of the brain organoid model that OrganoTherapeutics uses to develop new therapeutics against Parkinson’s disease. Hence, having available a broad spectrum of these stem cell lines from different individuals, with different PD associated mutations and different genetic backgrounds is a key prerequisite for the success of this endeavor. The newly concluded alliance between Juntendo University in Tokyo and OrganoTherapeutics, now allows OrganoTherapeutics to make use of various stem cell lines that have been generated at Juntendo. ➡ OrganoTherapeutics CEO, Jens Schwamborn mentions “Juntendo University is a world leading institution in PD research. Partnering with such a powerhouse is an immense honor for us. Further, the cell lines that we get from Juntendo are extremely valuable for us and will certainly accelerate our therapeutics developments. We are very grateful for this important collaboration.” ➡ Chair and Professor of Juntendo University, Nobutaka Hattori mentions “We work with collaborators around the world to bring better treatments to patients with Parkinson's disease. Through this partnership with OrganoTherapeutics, a frontrunner in the development of therapeutics using iPSC-derived organoids, we are excited about the development of new therapies.” About Juntendo University: Juntendo University was founded in 1838 as the oldest Western medical school in Japan. The Department of Neurology at Juntendo University has focused on the treatment and research of Parkinson's disease for many years. They have made many research contributions, including the discovery of three PARK PD-related genes, a blood-based PD diagnostic method, and studies using patient-derived iPS cells. Their goal is to provide more optimal treatment for all patients through the development of these studies. About OrganoTherapeutics: OrganoTherapeutics (OT) is a spin-off biotech company from the University of Luxembourg and the Luxembourg center for Systems Biomedicine. Founded in 2019, OT aims at further developing brain organoid systems and using them for therapeutics development in Parkinson’s disease. The OT approach is based on phenotypic screening and AI based data analysis. The company is well embedded in the Luxembourgish research landscape, collaborates with leading institutions through European networks and has established partnerships with universities, biotech and pharma companies in Europe, Japan and the US. #collaboration #parkinsonsdisease #therapeutics
OrganoTherapeutics’ Post
More Relevant Posts
-
A complete study of 𝐒𝐭𝐞𝐦 𝐂𝐞𝐥𝐥 𝐓𝐡𝐞𝐫𝐚𝐩𝐲 [PDF Data] 𝐃𝐨𝐰𝐧𝐥𝐨𝐚𝐝 𝐅𝐫𝐞𝐞 𝐏𝐃𝐅 𝐂𝐨𝐩𝐲: https://lnkd.in/dHR3TQZE (Please prefer the corporate mail ID to get the highest priority) The new research study consists of an industry trend analysis of the market. The new research study consists of industry trends, pricing analysis, patent analysis, conference and webinar materials, key stakeholders, and buying behaviour in the market. Market growth is driven mainly by factors such as robust funding for stem cell research, an increase in the number of cell therapy production facilities that have GMP certification, and growth in the number of clinical trials associated with stem cell therapies. The surge in the expanding number of clinical trials is further triggered by the COVID-19 pandemic, with a vast number of clinical trials studying the potential of stem cell therapies in the treatment of SARS-CoV-2 infection. 𝑺𝒕𝒆𝒎 𝑪𝒆𝒍𝒍 𝑻𝒉𝒆𝒓𝒂𝒑𝒚 𝑴𝒂𝒓𝒌𝒆𝒕 𝑫𝒚𝒏𝒂𝒎𝒊𝒄𝒔 𝑫𝒓𝒊𝒗𝒆𝒓: 𝑨𝒗𝒂𝒊𝒍𝒂𝒃𝒊𝒍𝒊𝒕𝒚 𝒐𝒇 𝒇𝒖𝒏𝒅𝒊𝒏𝒈 𝒇𝒐𝒓 𝒔𝒕𝒆𝒎 𝒄𝒆𝒍𝒍 𝒓𝒆𝒔𝒆𝒂𝒓𝒄𝒉 Stem cell research funding has strengthened in the past few years, attributed to the need for novel therapeutic interventions in patients with cardiovascular, neurological, and autoimmune disorders. The NIH funding for stem cell research reached its highest peak in 2018 and has shown a year-on-year growth of more than 5% in the stem cell research funding each year. This trend is expected to continue with the increase in funding initiatives announced in 2021. For instance, in September 2021, the California Institute for Regenerative Medicine granted a grant of USD 31 million to Stanford Research for launching first-in-human trials of stem cells in the treatment of heart failure, stroke, and spinal cord tumours. 𝑵𝒐𝒓𝒕𝒉 𝑨𝒎𝒆𝒓𝒊𝒄𝒂 𝒘𝒂𝒔 𝒕𝒉𝒆 𝒍𝒂𝒓𝒈𝒆𝒔𝒕 𝒓𝒆𝒈𝒊𝒐𝒏 𝒇𝒐𝒓 𝒔𝒕𝒆𝒎 𝒄𝒆𝒍𝒍 𝒕𝒉𝒆𝒓𝒂𝒑𝒚 𝒎𝒂𝒓𝒌𝒆𝒕. Geographically, the market is segmented into North America, Europe, Asia Pacific, and RoW (the rest of the world). In 2022, the global market was dominated by the North America region, and this dominance is anticipated to continue throughout the forecast period. The US has emerged as the key revenue contributor to this region, with robust public-private funding to develop novel stem cell therapy products. Prominent players in the stem cell therapy market include Smith+Nephew (UK), MEDIPOST Co., Ltd. (South Korea), Anterogen.Co., Ltd. (South Korea), CORESTEM (South Korea), Pharmicell Inc. (South Korea), NuVasive, Inc. (US), RTI Surgical (US), AlloSource (US), JCR Pharmaceuticals Co., Ltd. (Japan), Takeda Pharmaceutical Company Limited (Japan), Holostem Terapie Avanzate Srl (Italy), Orthofix (US), Regrow Biosciences Pvt Ltd. (India), and STEMPEUTICS RESEARCH PVT LTD. (India).
To view or add a comment, sign in
-
-
A Complete Guide of " 𝐒𝐭𝐞𝐦 𝐂𝐞𝐥𝐥 𝐓𝐡𝐞𝐫𝐚𝐩𝐲 " [ PDF Guide] 𝐃𝐨𝐰𝐧𝐥𝐨𝐚𝐝 𝐅𝐫𝐞𝐞 𝐒𝐚𝐦𝐩𝐥𝐞 𝐑𝐞𝐩𝐨𝐫𝐭 𝐇𝐞𝐫𝐞: https://lnkd.in/dQDRwEJ6 The new research study consists of industry trends, pricing analysis, patent analysis, conference and webinar materials, key stakeholders, and buying behaviour in the market. Market growth is driven mainly by factors such as rise in funding for stem cell research, upsurge in the number of cell therapy production facilities that have GMP certification, and increased in the number of clinical trials associated with stem cell therapies. 𝐃𝐫𝐢𝐯𝐞𝐫: 𝐈𝐧𝐜𝐫𝐞𝐚𝐬𝐞𝐝 𝐟𝐮𝐧𝐝𝐢𝐧𝐠 𝐟𝐨𝐫 𝐬𝐭𝐞𝐦 𝐜𝐞𝐥𝐥 𝐫𝐞𝐬𝐞𝐚𝐫𝐜𝐡 Government initiatives and funding mechanisms exert a pivotal influence on the dynamics of the market. Favorable regulatory frameworks and financial support from governmental bodies play a catalytic role in fostering an environment conducive to research and development in stem cell therapies. The need for newer and better therapies for the treatment of autoimmune, neurological, and cardiovascular diseases has resulted in an overall increase in research activities and the availability of funding for cell-based research. In December 2022, Alpha Stem Cell Clinic received an USD 8 million, five-year grant from the California Institute of Regenerative Medicine (CIRM) for the study of the use of self-renewing cells to treat human disease. Furthermore, in November 2019, the Australian government released a 10-year roadmap for stem cell research in Australia—The Stem Cell Therapies Mission. The initiative would provide USD 102 million (AUD 150 million) under the Medical Research Future Fund (MRFF) to support stem cell research to deliver new therapies. The infusion of government funding not only accelerates the pace of clinical trials but also facilitates the translation of research findings into viable therapeutic applications. As a result, the stem cell therapy industry is experiencing a positive ripple effect, with increased investments leading to expanded capabilities, enhanced infrastructure, and ultimately contributing to the overall growth and maturation of the industry.
To view or add a comment, sign in
-
-
Bayer Breakthrough in Parkinson's Stem Cell Therapy! Exciting news from the medical world as Bayer announces a significant leap in the battle against Parkinson's disease. The experimental stem cell therapy developed by its U.S. subsidiary, BlueRock Therapeutics has shown promising results in easing Parkinson's symptoms during an early 12-patient trial. 🔍 Key Highlights: - 12 patients underwent the trial: seven on a high dose and five on a lower dose. - High dose participants reported an average of 2.16 hours longer with well-controlled symptoms per day, while the time of worsening symptoms was cut by 1.91 hours. - Lower dose participants experienced an average of 0.72 hours longer with controlled symptoms and 0.75 hours less of worsening symptoms. - No major safety issues were encountered. The treatment was well-tolerated. The therapy involves transforming human pluripotent embryonic stem cells into dopamine-producing nerve cells, which are then implanted into the brain. The results were shared at the International Congress of Parkinson's Disease and Movement Disorders in Copenhagen, Denmark. Bayer plans to move forward with testing in the second of three stages, with patient enrolment set to begin in the first half of 2024. Bayer's advancements in this area provide hope to over 10 million Parkinson's patients worldwide. As research progresses, we look forward to more breakthroughs in the quest for a cure. https://lnkd.in/eZMTfVpZ #ParkinsonsResearch #StemCellTherapy #MedicalBreakthroughs #BayerUpdates
To view or add a comment, sign in
-
(Pluripotent) Stem Cells | iPS Cells | ES Cells | Early Discovery | Cell Therapy | Regenerative Medicine | Precision Medicine | Drug Screening Platform(s) | Aging & Longevity | 🇨🇦 in 🇺🇸
Stem Cell Therapy Landscape: Last Episode Next series: Stem cells and Cultured meat The journey of stem cells from research labs to bedside treatments is a complex one, riddled with intricate technical challenges. A primary concern is the transition from academic bench protocols, which are often manual and small-scale, to robust, industrial-scale systems that adhere to Good Manufacturing Practices (GMP). This shift demands not only scalability but also a consistent and sustainable approach to ensure the quality and efficacy of the therapies. Innovative techniques, such as 3D mid-to-large scale bioprocessing, are being explored to produce human iPSC-derived cortical neurons more efficiently. These processes aim to enhance cell yield, purity, and functionality, which are crucial for therapeutic applications. Furthermore, the integration of AI and machine learning offers promising avenues to streamline and optimize the iPSC manufacturing process, from cell sourcing to differentiation and quality control. However, with these advancements come challenges. Ensuring that these high-tech processes maintain the integrity and safety of the stem cells, while also being scalable for clinical demands, is a delicate balance. As the field progresses, it's imperative to address these technical hurdles head-on, laying a solid foundation for the successful clinical translation of stem cell therapies. https://lnkd.in/gX2pYsXT
Autologous Induced Pluripotent Stem Cell–Based Cell Therapies: Promise, Progress, and Challenges
currentprotocols.onlinelibrary.wiley.com
To view or add a comment, sign in
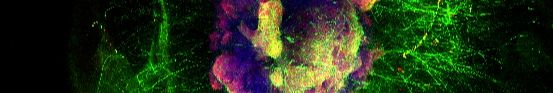
Congrats OT