How to Make Dilutions and Serial Dilutions
Dilutions
Dilutions
Making dilutions is a common part of science projects in many areas of science including chemistry, biology, medicine, cooking, and environmental sciences. This video and written tutorial will guide you through how to make a dilution and how to calculate the dilution factor. How to make serial dilutions is also explained.
If you need help making a stock solution to dilute, check out our How to Make Solutions for Chemistry and Biology Experiments tutorial.
Dilution
What is a dilution?
Dilution is the process of making a solution or scientific sample less concentrated by adding more solvent. For example, if you have a glass of lemonade but it is too sour, you might add more water to it to make a diluted, less sour glass of lemonade. In this example the starting solution (sometimes called a stock solution), is the original lemonade, the solvent is the water you add, and the dilution is the less-sour glass of lemonade you end up with.
Make Dilution
How to make a dilution
Follow these five steps to make a dilution:
- Calculate the volumes needed. If you do not know them already, use the dilution factor equation to calculate the volume of the original solution (V1) and the volume of the dilution you are making (V2).
- Take safety precautions. If you are using hazardous materials or need to keep your dilutions sterile (germ-free), you may need to use some safety equipment. The most common safety equipment for making dilutions in student science projects are:
- Latex or nitrile gloves: used to keep germs from your hands out of your solutions and most chemicals off of your skin. If you are using harsh chemicals, like a very strong acid or base, you may need to use chemical-resistant gloves. This is not common for most science projects.
- Chemical splash goggles or glasses: used to keep chemicals from getting in your eyes. If the chemicals you are using may irritate your eyes, these are a good idea.
- Measure the stock solution. Use a measuring cup, graduated cylinder, or pipette to measure out the volume of the stock solution (V1) from step one. Which you use will depend on the type of science project. A cooking science project may use a measuring cup. A chemistry project is likely to use a graduated cylinder or pipette for more accuracy.
- Add the solvent. Pour the stock solution into a graduated cylinder or other measuring container large enough to hold the final volume of the dilution. Slowly, mixing thoroughly as you go, add solvent until you reach the desired volume for the dilution (V2).
- Label and store the dilution. If needed, transfer your solution to a storage container. Immediately label the container with the name of the dilution, the concentration, and the date it was made.
Factor Equation
What is the dilution factor equation?
The dilution factor equation describes the relationship between the volume and concentration of the starting solution (sometimes called a stock solution) and the volume and concentration of the dilution you are preparing. It is written as:
Where:
- C1 = Concentration of the original/stock solution
- V1 = Volume of the original solution you will use to make your dilution
- C2 = Desired concentration of the diluted solution
- V2 = Volume of the diluted solution you want to make
If you know three out of the four parts of the dilution factor equation, you can use re-arrange the equation to calculate the fourth part.
| To calculate | Re-arranged dilution factor equation |
|---|---|
| Volume of the starting solution | |
| Volume of the dilution | |
| Concentration of the starting solution | |
| Concentration of the dilution |
The How to Dilute a Solution video takes you through several sample calculations.
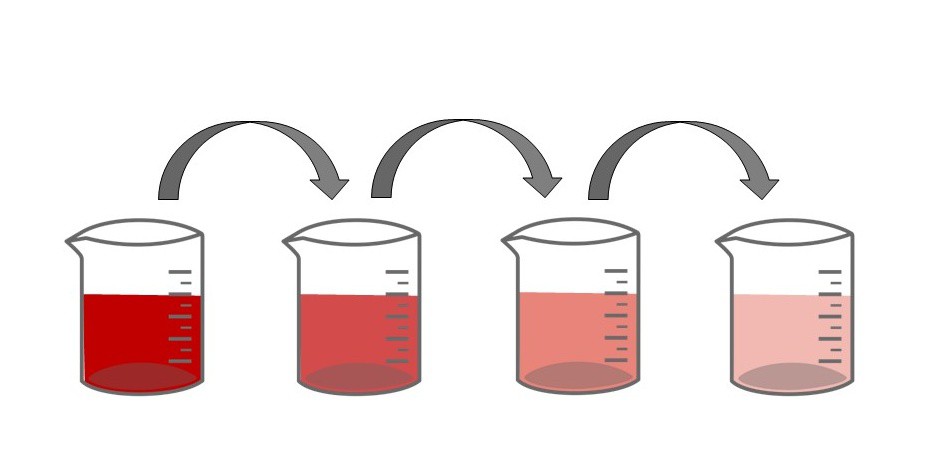
Serial Dilutions
How to make a serial dilution
A serial dilution is a stepwise series of dilutions. Each dilution in the series is used to make the next dilution until you have a whole set of dilutions with different concentrations.
Here is an example of a serial dilution.
| Solution | Concentration | Made with |
|---|---|---|
| Stock solution | 100% v/v | Pure lemon juice |
| Dilution #1 | 50% v/v | 50 mL of stock solution + 50 mL of water |
| Dilution #2 | 25% v/v | 50 mL of dilution #1 + 50 mL of water |
| Dilution #3 | 12.5% v/v | 50 mL of dilution #2 + 50 mL of water |
| Dilution #4 | 6.25% v/v | 50 mL of dilution #3 + 50 mL of water |