Answer
334.2k+ views
Hint: Sodium hydrogen pyro phosphite is an inorganic compound in which the elements present are oxygen, hydrogen, sodium, and phosphorus, so its formula is $N{{a}_{2}}{{H}_{2}}{{P}_{2}}{{O}_{7}}$. If the hydrogen atom is not attached to an electronegative atom then it is a non-acidic hydrogen atom.
Complete answer:
Disodium dihydrogen diphosphate is an IUPAC name of Sodium dihydrogen pyrophosphate which is an inorganic compound in which the elements present are oxygen, hydrogen, sodium, and phosphorus. The formula of Sodium hydrogen pyro phosphite is $N{{a}_{2}}{{H}_{2}}{{P}_{2}}{{O}_{7}}$.
Let us see the structure of the compound.
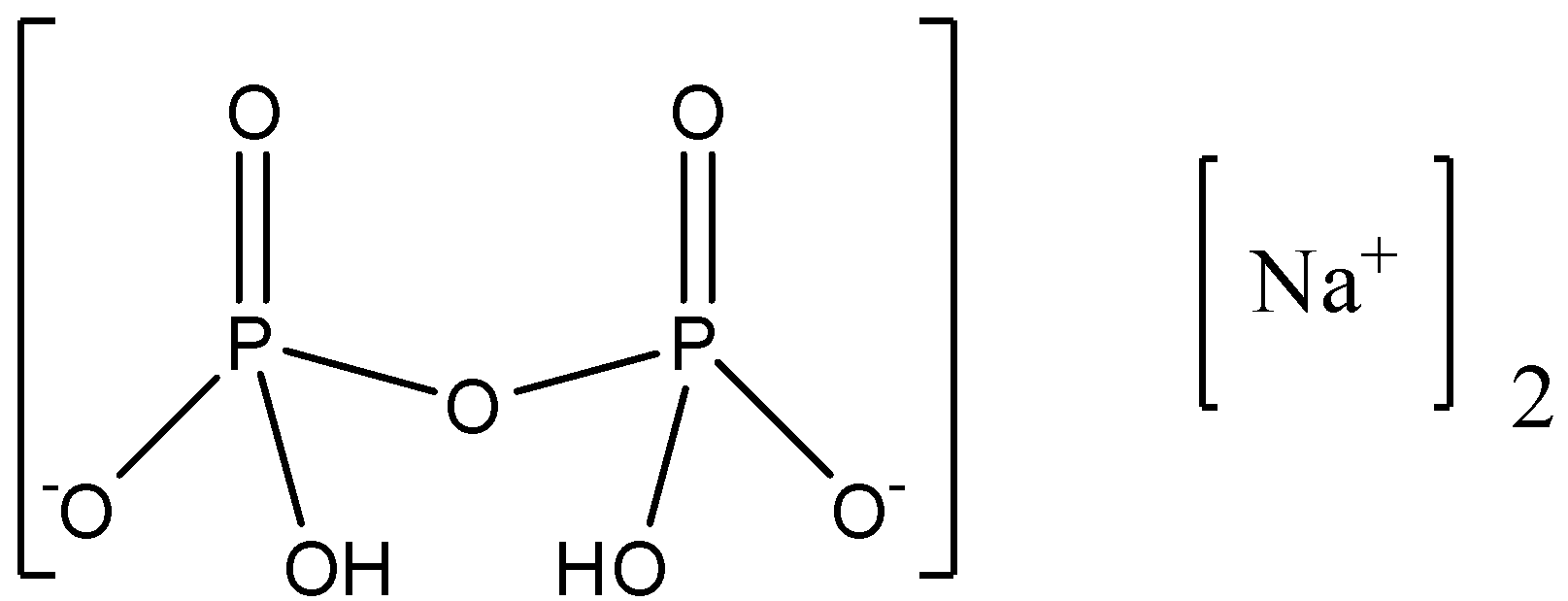
If the hydrogen atom is not attached to an electronegative atom then it is a non-acidic hydrogen atom. As we can see that there are two hydrogen atoms in the compound and both the hydrogen atoms are attached to the oxygen atom, an oxygen atom is electronegative, so both the hydrogen atoms are acidic hydrogen.
Therefore, the number of non-acidic hydrogen ‘x’ is 0.
From the formula we can see that the number of oxygen atoms are 7, therefore, ‘y ‘ is 7.
Now, we have to find the oxidation number of phosphorus in the compound. The oxidation state is calculated as follows:
$N{{a}_{2}}{{H}_{2}}{{P}_{2}}{{O}_{7}}$
$2\text{ x (+1) + 2 x}\text{ (+1) + 2}x\text{ + 7 x (-2) = 0}$
$\text{2 + 2 + 2}x\text{ - 14 = 0}$
$x=+5$
Therefore, the value of ‘z’ is +5.
Now, we have to find the value of (x+y-z), so by putting the values, we get:
$(x+y-z)=(0+7-(+5))$
$(x+y-z)=7-5=2$
So, the answer is 2.
Therefore, the correct answer is an option (d).
Note:
In the compound, if the hydrogen atom was directly attached to the phosphorus atom then, it would be counted as a non-acidic hydrogen atom. While calculating the oxidation state, the oxidation number of sodium is taken +1 because it is an alkali metal
Complete answer:
Disodium dihydrogen diphosphate is an IUPAC name of Sodium dihydrogen pyrophosphate which is an inorganic compound in which the elements present are oxygen, hydrogen, sodium, and phosphorus. The formula of Sodium hydrogen pyro phosphite is $N{{a}_{2}}{{H}_{2}}{{P}_{2}}{{O}_{7}}$.
Let us see the structure of the compound.

If the hydrogen atom is not attached to an electronegative atom then it is a non-acidic hydrogen atom. As we can see that there are two hydrogen atoms in the compound and both the hydrogen atoms are attached to the oxygen atom, an oxygen atom is electronegative, so both the hydrogen atoms are acidic hydrogen.
Therefore, the number of non-acidic hydrogen ‘x’ is 0.
From the formula we can see that the number of oxygen atoms are 7, therefore, ‘y ‘ is 7.
Now, we have to find the oxidation number of phosphorus in the compound. The oxidation state is calculated as follows:
$N{{a}_{2}}{{H}_{2}}{{P}_{2}}{{O}_{7}}$
$2\text{ x (+1) + 2 x}\text{ (+1) + 2}x\text{ + 7 x (-2) = 0}$
$\text{2 + 2 + 2}x\text{ - 14 = 0}$
$x=+5$
Therefore, the value of ‘z’ is +5.
Now, we have to find the value of (x+y-z), so by putting the values, we get:
$(x+y-z)=(0+7-(+5))$
$(x+y-z)=7-5=2$
So, the answer is 2.
Therefore, the correct answer is an option (d).
Note:
In the compound, if the hydrogen atom was directly attached to the phosphorus atom then, it would be counted as a non-acidic hydrogen atom. While calculating the oxidation state, the oxidation number of sodium is taken +1 because it is an alkali metal
Recently Updated Pages
Basicity of sulphurous acid and sulphuric acid are

Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

What is the stopping potential when the metal with class 12 physics JEE_Main

The momentum of a photon is 2 times 10 16gm cmsec Its class 12 physics JEE_Main

How do you arrange NH4 + BF3 H2O C2H2 in increasing class 11 chemistry CBSE

Is H mCT and q mCT the same thing If so which is more class 11 chemistry CBSE

Trending doubts
Difference Between Plant Cell and Animal Cell

Difference between Prokaryotic cell and Eukaryotic class 11 biology CBSE

Fill the blanks with the suitable prepositions 1 The class 9 english CBSE

Change the following sentences into negative and interrogative class 10 english CBSE

How many millions make a billion class 6 maths CBSE

Fill the blanks with proper collective nouns 1 A of class 10 english CBSE

Give 10 examples for herbs , shrubs , climbers , creepers

What organs are located on the left side of your body class 11 biology CBSE

What is BLO What is the full form of BLO class 8 social science CBSE



